- Record: found
- Abstract: found
- Article: found
SGLT2 Inhibition by Empagliflozin Promotes Fat Utilization and Browning and Attenuates Inflammation and Insulin Resistance by Polarizing M2 Macrophages in Diet-induced Obese Mice
Read this article at
Abstract
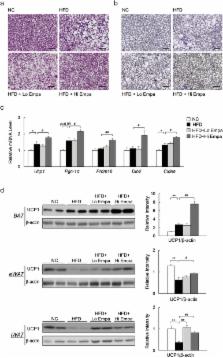
Sodium-glucose cotransporter (SGLT) 2 inhibitors increase urinary glucose excretion (UGE), leading to blood glucose reductions and weight loss. However, the impacts of SGLT2 inhibition on energy homeostasis and obesity-induced insulin resistance are less well known. Here, we show that empagliflozin, a SGLT2 inhibitor, enhanced energy expenditure and attenuated inflammation and insulin resistance in high-fat-diet-induced obese (DIO) mice. C57BL/6J mice were pair-fed a high-fat diet (HFD) or a HFD with empagliflozin for 16 weeks. Empagliflozin administration increased UGE in the DIO mice, whereas it suppressed HFD-induced weight gain, insulin resistance, and hepatic steatosis. Moreover, empagliflozin shifted energy metabolism towards fat utilization, elevated AMP-activated protein kinase and acetyl-CoA carbolxylase phosphorylation in skeletal muscle, and increased hepatic and plasma fibroblast growth factor 21 levels. Importantly, empagliflozin increased energy expenditure, heat production, and the expression of uncoupling protein 1 in brown fat and in inguinal and epididymal white adipose tissue (WAT). Furthermore, empagliflozin reduced M1-polarized macrophage accumulation while inducing the anti-inflammatory M2 phenotype of macrophages within WAT and liver, lowering plasma TNFα levels and attenuating obesity-related chronic inflammation. Thus, empagliflozin suppressed weight gain by enhancing fat utilization and browning and attenuated obesity-induced inflammation and insulin resistance by polarizing M2 macrophages in WAT and liver.
Highlights
-
•
SGLT2 inhibition by empagliflozin enhances energy expenditure and thermogenesis by promoting the browning of fat.
-
•
Empagliflozin enhances AMPKα and ACC phosphorylation in skeletal muscle and increases hepatic and plasma levels of FGF21.
-
•
Empagliflozin attenuates obesity-induced inflammation and insulin resistance by polarizing M2 macrophages in fat and liver.
Sodium-glucose cotransporter 2 (SGLT2) inhibition by empagliflozin increases glucosuria, thereby reducing hyperglycaemia and weight with pleiotropic effects in obesity: (1) Empagliflozin promotes fat utilization by activating AMPK in skeletal muscle. (2) Empagliflozin increases fibroblast growth factor (FGF) 21 levels in blood and promotes browning of adipose tissue, thereby increasing thermogenesis and energy expenditure. (3) Empagliflozin reduces M1-polarized macrophage accumulation while inducing the anti-inflammatory M2 phenotype of macrophages within fat and liver, attenuating obesity-induced inflammation and insulin resistance.
Related collections
Most cited references17
- Record: found
- Abstract: found
- Article: not found
Macrophage-specific PPARgamma controls alternative activation and improves insulin resistance.
- Record: found
- Abstract: found
- Article: not found
Group 2 innate lymphoid cells promote beiging of white adipose tissue and limit obesity.
- Record: found
- Abstract: found
- Article: not found