- Record: found
- Abstract: found
- Article: found
Uncontained spread of Fusarium wilt of banana threatens African food security
review-article
Anouk C. van Westerhoven
1
,
2 ,
Harold J. G. Meijer
3 ,
Michael F. Seidl
2
,
*
,
,
Gert H. J. Kema
1
,
*
,
22 September 2022
Read this article at
There is no author summary for this article yet. Authors can add summaries to their articles on ScienceOpen to make them more accessible to a non-specialist audience.
Abstract
Why is banana among the most vulnerable crops?
Banana is the most popular fruit worldwide [1] and a major staple food in tropical
and subtropical regions where the majority of bananas is produced (
Fig 1
) [2]. The importance of banana for food security is particularly relevant for East
Africa (Burundi, Congo, Rwanda, Tanzania, and Uganda). Here, the East African Highland
bananas (EAHBs) are the crucial cash crops and staple food for millions of people
with the world’s highest per capita banana consumption of 400 to 600 kg [3]. Throughout
this region, banana cultivation is embedded in complex mixed cropping systems by numerous
small-scale farmers and households [4]. Most edible bananas are seedless parthenocarpic
diploids and triploid hybrids derived from the wild banana species Musa acuminata
and Musa balbisiana [5]. Although the wild, seeded bananas are genetically very diverse
[5], the domestication of seedless and hence edible banana varieties resulted in a
genetic bottleneck that limits genetic variation [5]. On the local market, different
clonal banana varieties are sold, in contrast to the global banana trade that is dominated
by clonal Cavendish varieties [6]. These large banana monocultures are extremely vulnerable
to numerous diseases [7].
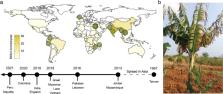
10.1371/journal.ppat.1010769.g001
Fig 1
Uncontained spread of Fusarium wilt in banana caused by Fusarium odoratissimum TR4.
(A) Banana is a major food crop in tropical and subtropical regions, especially in
sub-Saharan Africa. In most major banana-producing regions, TR4 incursions have been
reported (green dots), and TR4 is spreading globally from its Asian center of origin
to other banana-growing regions [8,19–27]. The colors of the countries on the global
map indicate banana production in million tonnes per year. Map downloaded from Natural
Earth Data; https://www.naturalearthdata.com. (B) Cavendish banana plant in Mozambique
showing external FWB symptoms, caused by TR4 [27].
A major concern for banana production is Fusarium wilt, a devastating vascular disease
that withers banana plants (
Fig 1
). It is caused by soil-borne fungi belonging to the Fusarium oxysporum species complex.
Strains that are able to infect banana were known as F. oxysporum f.sp. cubense (Foc),
despite their well-known diversity [8]. Recently, genotyping analyses confirmed several
genetically distinct Foc lineages that were consequently recognized as individual
Fusarium species [9]. For instance, the Foc Race 1 strains, which drove a major epidemic
of Fusarium wilt of banana (FWB) that almost eradicated the Gros Michel variety dominating
the banana trade up to the 1960s in the last century [8], actually comprise a suite
of different Fusarium species [9]. Eventually, the resistant Cavendish clones saved
the industry, and nowadays, 98% of the export market depends on them [6]. However,
already in 1967, FWB affected Cavendish in Taiwan [8] (
Fig 1
). The causal Fusarium strain, referred to as Tropical Race 4 (TR4), has recently
been described as the new species, Fusarium odoratissimum [9]. This modified nomenclature
of FWB causing Fusarium spp. raised some controversy [10], and therefore awaits additional
conclusive data. Most experts, however, agree that TR4 is a clonal lineage and genetically
so dissimilar from other banana infecting Fusarium spp. that it is justifiably recognized
as a new species. Next to Cavendish cultivars, TR4 affects a wide range of banana
germplasm, including locally important varieties, such as the aforementioned EAHBs
[11]. The latter are essential for food security in the African Great Lakes Region
where banana is a major staple crop that already suffers from manifold other pests
and diseases, such as nematodes, weevils, Xanthomonas bacterial wilt, and black leaf
streak disease, also known as Black Sigatoka.
How to respond to a Fusarium wilt incursion?
No commercially available banana variety is resistant to TR4, and consequently, surveillance
and disease management are currently the only strategies to control its further dissemination.
Traditionally, TR4 incursions were identified based on visual wilting symptoms in
Cavendish plants in combination with vegative compatability group (VCG) testing. During
this procedure, a nitrate nonutilizing (nit) mutant of a fungal isolate is grown on
a Petri dish with known tester strains to assess the ability to form a stable heterokaryon
and hence score its compatibility. Such strains are grouped into the same VCG, and
TR4 is categorized as VCG 01213 (sometimes also referred to as VCG 01216 [12]). However,
this procedure is time-consuming and does not yield reliable results [13]. To curb
these disadvantages, a diagnostic PCR was developed and commercialized [14]. Later,
other diagnostics became available [15,16], including a fast and easy LAMP test, targeting
a different genomic region, which enables rapid identification of TR4 even under field
conditions [17]. However, sooner or later, every diagnostic will retrieve false positives,
such as by a non-TR4 strain that nevertheless tests positive [18]. Therefore, multiple
diagnostics should be used that target different genomic areas for confirmatory reasons.
Furthermore, they require continuous monitoring of reliability and consequently updates
once false positives are observed, and they should only be evaluated with the biological
material for which they were developed. In addition to quick molecular diagnostics,
sequencing technologies have made VCG testing redundant as they provide the required
resolution to determine the homogeneity and phylogeography of TR4 dissemination. Notably,
the genome sequences of various TR4-isolates sampled from independent incursions worldwide
reveal very little genetic variation, suggesting its clonal origin ([19]; S1 Dataset).
The improved identification and tracing of TR4 are expected to enable rapid implementation
and refinement of containment strategies. Nevertheless, FWB caused by TR4 is swiftly
spreading across many banana-growing countries worldwide (
Fig 1
).
Are there options for continued banana production after a Fusarium incursion?
Upon the first reports in Taiwan [8], TR4 disseminated across South East Asia [19].
In 2013, the first incursion outside South East Asia was reported in Jordan [20] (
Fig 1
). Since then, 12 incursions followed in the Middle East [21], the Indian subcontinent
[22], Africa [23,24], and most recently in Latin America [25–27] (
Fig 1
).
The arrival of TR4 in Mozambique in 2013 is highly significant due to the importance
of bananas as a staple crop in sub-Saharan Africa. Presumably, the TR4 incursion was
restricted to 2 commercial plantations in the North of the country [28]. The plantations
were placed under quarantine [28] but production was continued, partly with GCTCV218,
a less susceptible Cavendish mutant [29]. During surveys in 2015, no suspicious wilting
symptoms were detected outside the farms; hence, TR4 was declared to be under control
[28]. However, recently, wilting symptoms were observed outside the farm boundaries
and subsequent analyses confirmed the dissemination of TR4 to other distant locations
(
Fig 1
) [30]. A comparison of 5 fungal strains, isolated from FWB symptomatic banana plants
at various locations, to the TR4 II5 reference isolate clearly confirmed TR4 as the
causal organism [30]. The analyzed isolates show little genetic diversity [30], suggesting
that local transmission occurs through a single clonal lineage. However, the isolates
could not be linked to other worldwide reported TR4 incursions since sequencing data
typically only include isolates from the first official disease reports that lack
sampling depth to address local diversity and dissemination ([30]; S1 Dataset). Consequently,
comparative analyses of the genomes from new versus previous incursions enables only
provisional associations. The accumulation of unique genetic variation across TR4
strains in Mozambique suggests an extended time of local spread [30]. We, therefore,
can neither robustly link the origin of TR4 in Mozambique with incursions in other
countries, nor declare the newly discovered TR4 strains as independent novel incursions.
We even cannot link them with the 2 initially infested farms because there are no
publicly available sequencing data from the strains originally identified at these
farms [30]. However, the proximity of the sampling sites to these farms and the applied
disease management practices strongly suggest that TR4 was not successfully contained.
Moreover, TR4 was recently reported on Mayotte, an island in the Indian Ocean approximately
700 km from the infested farms in Mozambique [24]. Again, the origin of this incursion
is unknown due to lacking sequencing data. This underscores the importance of sequencing
efforts and rapid data sharing to unveil whether disease management efforts were effective
[31].
Taken together, it is very likely that the spread of TR4 in Mozambique was not stopped
by cultivating less susceptible Cavendish (GCTCV) somaclones. On the contrary, it
is conceivable that this management practice contributed to the further dissemination
of TR4. Reduced susceptibility of alternative banana germplasm seems inadequate to
manage FWB. Complete resistance is required as shown by the Cavendish varieties that
are globally cultivated on Race 1–infested soils for over 70 years without any decline
of resistance [8]. Any level of resistance to TR4 that does not meet this standard
is insufficient and can contribute to further dissemination of TR4.
Can FWB caused by TR4 be stopped?
FWB management strategies currently aim to prevent the spread of TR4 by focusing on
the use of clean planting material and machinery and the quarantining of infested
farms [13,28,32]. However, the ongoing global spread shows that FWB successfully disseminates
despite extensive prevention strategies (
Fig 1
). The case study of Mozambique is not the only example of the challenges associated
with the containment of TR4 following an initial incursion. The spread of TR4 to Colombia
is yet another case. Upon the first report of TR4 in the Guajira department in 2019
[25], Colombia declared a state of emergence entailing sanitary control measures as
well as aerial and on-the-ground inspections as part of the containment strategy [32].
Nevertheless, in 2021, TR4 also appeared in the neighboring Magdalena department [27],
demonstrating the spread of TR4 irrespective of the implemented containment strategies.
A recent incursion in Peru [26] illustrates ongoing local and global spread but is
considered to be independent of the presence of TR4 in Colombia [27]. However, this
can only be concluded after sufficient sampling in Colombia. These collective data
underscore the failure of contemporary management strategies for TR4. The uncontained
spread that drives the TR4 pandemic is reminiscent of the previous dissemination of
Race 1 strains [7,8] and puts regions at risk that rely on bananas. Notably, the documented
spread of TR4 largely involves commercial banana farms. However, the unsuccessful
disease management at these large farms raises the concern for small-scale farms that
dominate African banana production. Smallholders are frequently disconnected from
extension and cannot afford or are less skilled in disease and pest management [4].
Hence, the occurrence of TR4 outside major farms is largely unattended, facilitates
its spread, and directly threatens income and food security.
The risks posed by emerging and spreading plant pathogens are increasingly recognized
[33]. Like FWB, various plant pathogens affect important food crops. For example,
wheat blast, caused by Magnaporthe oryzae pathotype Triticum, originates from Brazil
[34] but destroyed 50% of wheat crops after it emerged in Bangladesh in 2016 and was
recently detected in Zambia [35]. Next to their impact on agriculture, invasive fungal
diseases on plants and animals can also endanger natural ecosystems [36]. For example,
the damage to forests by ash dieback (caused by Hymenoscyphus fraxineus) in Europe
affects biodiversity and accounts for losses in fixed CO2 [37]. Similarly, the fungal
pathogen Batrachochytrium dendrobatidis causes a significant decline in amphibian
populations [36]. Human factors such as increased international travel or environmental
and climate changes likely drive pathogens’ emergence, evolution, and dissemination
to novel geographic regions or ecological niches [33]. Often new incursions remain
unnoticed and once fungal pathogens are endemic, successful disease management is
basically unfeasible, as exemplified by the very few examples of successful eradication
[38,39]. Such cases often rely on fungicides and thorough eradication of host plants,
illustrating the importance of an accurate understanding of the host range of a pathogen
[39]. Effective and open science at local and global scales are indispensable to enable
a rapid and coordinated response to emerging and invasive fungal diseases [31]. TR4
continues to disseminate (
Fig 1
), irrespective of implemented strategies, and we observe that new incursions often
do not lead to effective and transparent responses and data sharing, which are required
to improve disease control. The recently reported uncontrolled dissemination of FWB
in Mozambique [30] is a serious threat to African food security and global banana
production. Now, nearly 10 years after its introduction to Africa, we call for radical
eradication strategies of TR4, along with proactive screening for resistance of African
banana germplasm and intensified breeding programs for this important staple crop.
Supporting information
S1 Dataset
Overview of the samples used in the study.
(XLSX)
Click here for additional data file.
Related collections
Most cited references35
- Record: found
- Abstract: found
- Article: not found
Emerging fungal threats to animal, plant and ecosystem health.
Matthew Fisher, Daniel Henk, Cheryl Briggs … (2012)
- Record: found
- Abstract: found
- Article: not found
Multidisciplinary perspectives on banana (Musa spp.) domestication.
Christophe Jenny, K. Tomekpe, Vincent Lebot … (2011)

- Record: found
- Abstract: found
- Article: found
Fusarium Wilt of Banana: Current Knowledge on Epidemiology and Research Needs Toward Sustainable Disease Management
Miguel Dita, Marcia Barquero, Daniel Heck … (2018)