- Record: found
- Abstract: found
- Article: not found
In silico identification of potential inhibitors of vital monkeypox virus proteins from FDA approved drugs

Read this article at
Abstract
Abstract
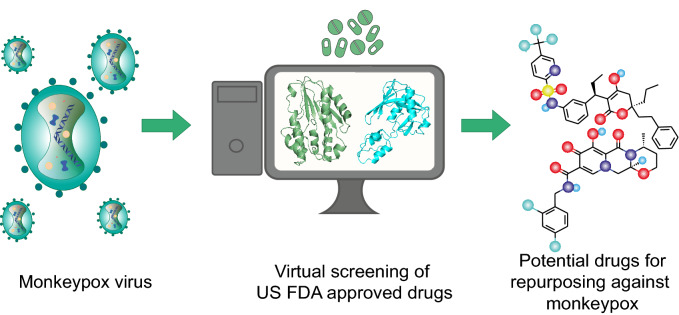
The World Health Organization (WHO) recently declared the monkeypox outbreak ‘A public health emergency of international concern’. The monkeypox virus belongs to the same Orthopoxvirus genus as smallpox. Although smallpox drugs are recommended for use against monkeypox, monkeypox-specific drugs are not yet available. Drug repurposing is a viable and efficient approach in the face of such an outbreak. Therefore, we present a computational drug repurposing study to identify the existing approved drugs which can be potential inhibitors of vital monkeypox virus proteins, thymidylate kinase and D9 decapping enzyme. The target protein structures of the monkeypox virus were modelled using the corresponding protein structures in the vaccinia virus. We identified four potential inhibitors namely, Tipranavir, Cefiderocol, Doxorubicin, and Dolutegravir as candidates for repurposing against monkeypox virus from a library of US FDA approved antiviral and antibiotic drugs using molecular docking and molecular dynamics simulations. The main goal of this in silico study is to identify potential inhibitors against monkeypox virus proteins that can be further experimentally validated for the discovery of novel therapeutic agents against monkeypox disease.
Related collections
Most cited references88
- Record: found
- Abstract: found
- Article: not found
AutoDock Vina: improving the speed and accuracy of docking with a new scoring function, efficient optimization, and multithreading.
- Record: found
- Abstract: not found
- Article: not found
GROMACS: High performance molecular simulations through multi-level parallelism from laptops to supercomputers
- Record: found
- Abstract: found
- Article: found