- Record: found
- Abstract: found
- Article: found
Dynamic actin cycling through mitochondrial subpopulations locally regulates the fission–fusion balance within mitochondrial networks
Read this article at
Abstract
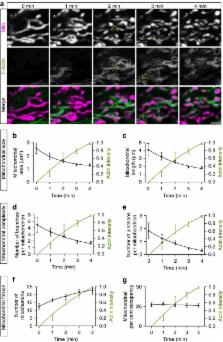
Mitochondria form interconnected networks that dynamically remodel in response to cellular needs. Using live-cell imaging, we investigate the role of the actin cytoskeleton in regulating mitochondrial fission and fusion. We identify cycling of actin filaments onto and off of subsets of cellular mitochondria. The association of actin filaments with mitochondrial subpopulations is transient; actin quickly disassembles, then reassembles around a distinct subpopulation, efficiently cycling through all cellular mitochondria within 14 min. The focal assembly of actin induces local, Drp1-dependent fragmentation of the mitochondrial network. On actin disassembly, fragmented mitochondria undergo rapid fusion, leading to regional recovery of the tubular mitochondrial network. Cycling requires dynamic actin polymerization and is blocked by inhibitors of both Arp2/3 and formins. We propose that cyclic assembly of actin onto mitochondria modulates the fission/fusion balance, promotes network remodelling and content mixing, and thus may serve as an essential mechanism regulating mitochondrial network homeostasis.
Abstract
 Mitochondria are dynamic organelles that can undergo fission and fusion. Here the
authors identify a novel pathway in which actin dynamically assembles in an Arp2/3-
and formin-dependent manner around a subset of cellular mitochondria, promoting localized
Drp1-dependent fission and impeding fusion.
Mitochondria are dynamic organelles that can undergo fission and fusion. Here the
authors identify a novel pathway in which actin dynamically assembles in an Arp2/3-
and formin-dependent manner around a subset of cellular mitochondria, promoting localized
Drp1-dependent fission and impeding fusion.
Related collections
Most cited references21
- Record: found
- Abstract: found
- Article: not found
Chemical inhibition of the mitochondrial division dynamin reveals its role in Bax/Bak-dependent mitochondrial outer membrane permeabilization.
- Record: found
- Abstract: found
- Article: not found
Mitochondrial dynamics and inheritance during cell division, development and disease.
- Record: found
- Abstract: found
- Article: not found