- Record: found
- Abstract: found
- Article: found
Creation of a long-acting nanoformulated dolutegravir
Read this article at
Abstract
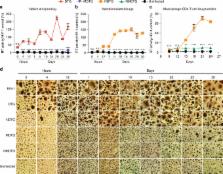
Potent antiretroviral activities and a barrier to viral resistance characterize the human immunodeficiency virus type one (HIV-1) integrase strand transfer inhibitor dolutegravir (DTG). Herein, a long-acting parenteral DTG was created through chemical modification to improve treatment outcomes. A hydrophobic and lipophilic modified DTG prodrug is encapsulated into poloxamer nanoformulations (NMDTG) and characterized by size, shape, polydispersity, and stability. Retained intracytoplasmic NMDTG particles release drug from macrophages and attenuate viral replication and spread of virus to CD4+ T cells. Pharmacokinetic tests in Balb/cJ mice show blood DTG levels at, or above, its inhibitory concentration 90 of 64 ng/mL for 56 days, and tissue DTG levels for 28 days. NMDTG protects humanized mice from parenteral challenge of the HIV-1 ADA strain for two weeks. These results are a first step towards producing a long-acting DTG for human use by affecting drug apparent half-life, cell and tissue drug penetration, and antiretroviral potency.
Abstract
Current ART for treatment of HIV-1 infection requires a strict daily regimen adherence. Herein, the authors report the manufacture and characterization of a nanoformulated dolutegravir prodrug with improved cell and tissue penetration, a remarkable apparent half-life and the potential for bimonthly drug administration.
Related collections
Most cited references58
- Record: found
- Abstract: found
- Article: not found
Efficient isolation and propagation of human immunodeficiency virus on recombinant colony-stimulating factor 1-treated monocytes
- Record: found
- Abstract: found
- Article: not found
Adherence to highly active antiretroviral therapy (HAART): a meta-analysis.
- Record: found
- Abstract: found
- Article: not found