- Record: found
- Abstract: found
- Article: found
Anchoring zero valence single atoms of nickel and iron on graphdiyne for hydrogen evolution
Read this article at
Abstract
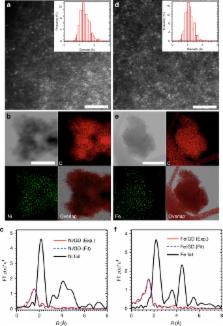
Electrocatalysis by atomic catalysts is a major focus of chemical and energy conversion effort. Although transition-metal-based bulk electrocatalysts for electrochemical application on energy conversion processes have been reported frequently, anchoring the stable transition-metal atoms (e.g. nickel and iron) still remains a practical challenge. Here we report a strategy for fabrication of ACs comprising only isolated nickel/iron atoms anchored on graphdiyne. Our findings identify the very narrow size distributions of both nickel (1.23 Å) and iron (1.02 Å), typical sizes of single-atom nickel and iron. The precision of this method motivates us to develop a general approach in the field of single-atom transition-metal catalysis. Such atomic catalysts have high catalytic activity and stability for hydrogen evolution reactions.
Abstract
Single atom catalysts provide the most efficient metal atoms usage and afford active site homogeneity, but surface attachment has proven challenging. Here, the authors use triple-bond-rich graphdiyne to anchor nickel/iron atoms and show high hydrogen evolution electrocatalysis activities.
Related collections
Most cited references41
- Record: found
- Abstract: not found
- Article: not found
First-principles calculations of the electronic structure and spectra of strongly correlated systems: theLDA+Umethod
- Record: found
- Abstract: found
- Article: not found
Water photolysis at 12.3% efficiency via perovskite photovoltaics and Earth-abundant catalysts.
- Record: found
- Abstract: not found
- Article: not found