- Record: found
- Abstract: found
- Article: found
Genetic Counseling in Kidney Disease: A Perspective
Read this article at
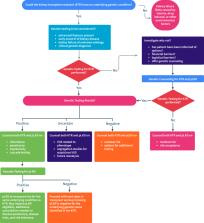
Abstract
As genetic testing is increasingly integrated into nephrology practice there is a growing need for partnership with genetic experts. Genetic counselors are ideally suited to fill this role. The value of genetic counseling is born out of the clinical value of genetic test results against the backdrop of the complexity of genetic testing. Genetic counselors who specialize in nephrology are trained to understand and explain the potential effects of genes on kidney disease, which can enable patients to make informed decisions about proceeding with genetic testing, navigating variants of uncertain significance, educating on extrarenal features of hereditary kidney disease, facilitating cascade testing, providing post-test education about testing results, and assisting with family planning. Genetic counselors can partner with the nephrologist and provide the knowledge needed to maximize the use of genetic testing for patients for nephrology consultation. Genetic counseling is more than an element or extension of genetic testing; it is a dynamic, shared conversation between the patient and the genetic counselor where concerns, sentiments, information, and education are exchanged, and value-based decision making is facilitated.
Related collections
Most cited references32
- Record: found
- Abstract: not found
- Article: not found
Atypical hemolytic-uremic syndrome.
- Record: found
- Abstract: found
- Article: not found