- Record: found
- Abstract: found
- Article: found
Complex roles of filamin-A mediated cytoskeleton network in cancer progression
Read this article at
Abstract
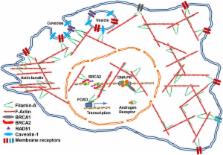
Filamin-A (FLNA), also called actin-binding protein 280 (ABP-280), was originally identified as a non-muscle actin binding protein, which organizes filamentous actin into orthogonal networks and stress fibers. Filamin-A also anchors various transmembrane proteins to the actin cytoskeleton and provides a scaffold for a wide range of cytoplasmic and nuclear signaling proteins. Intriguingly, several studies have revealed that filamin-A associates with multiple non-cytoskeletal proteins of diverse function and is involved in several unrelated pathways. Mutations and aberrant expression of filamin-A have been reported in human genetic diseases and several types of cancer. In this review, we discuss the implications of filamin-A in cancer progression, including metastasis and DNA damage response.
Related collections
Most cited references131
- Record: found
- Abstract: found
- Article: not found
Filamins as integrators of cell mechanics and signalling.
- Record: found
- Abstract: found
- Article: not found
Defining molecular profiles of poor outcome in patients with invasive bladder cancer using oligonucleotide microarrays.
- Record: found
- Abstract: found
- Article: not found