- Record: found
- Abstract: found
- Article: found
Recent advances in cerebral cavernous malformation research
Read this article at
Abstract
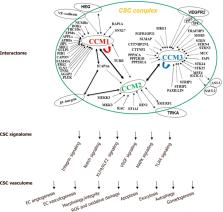
Cerebral cavernous malformations (CCM) are manifested by microvascular lesions characterized by leaky endothelial cells with minimal intervening parenchyma predominantly in the central nervous system predisposed to hemorrhagic stroke, resulting in focal neurological defects. Till date, three proteins are implicated in this condition: CCM1 (KRIT1), CCM2 (MGC4607), and CCM3 (PDCD10). These multi-domain proteins form a protein complex via CCM2 that function as a docking site for the CCM signaling complex, which modulates many signaling pathways. Defects in the formation of this signaling complex have been shown to affect a wide range of cellular processes including cell-cell contact stability, vascular angiogenesis, oxidative damage protection and multiple biogenic events. In this review we provide an update on recent advances in structure and function of these CCM proteins, especially focusing on the signaling cascades involved in CCM pathogenesis and the resultant CCM cellular phenotypes in the past decade.
Related collections
Most cited references156
- Record: found
- Abstract: found
- Article: not found
The Nrf2 cell defence pathway: Keap1-dependent and -independent mechanisms of regulation.

- Record: found
- Abstract: found
- Article: found
Antioxidant responses and cellular adjustments to oxidative stress
- Record: found
- Abstract: found
- Article: not found