- Record: found
- Abstract: found
- Article: found
Synthesis of trifluoromethyl-containing isoindolinones from tertiary enamides via a cascade radical addition and cyclization process†
research-article
3 July 2018
27 June 2018
Read this article at
There is no author summary for this article yet. Authors can add summaries to their articles on ScienceOpen to make them more accessible to a non-specialist audience.
Abstract
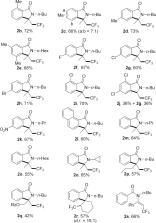
A radical trifluoromethylation reaction of tertiary enamides was investigated and trifluoromethyl-containing isoindolinones were prepared under mild conditions. Using TMSCF 3 as a radical source, PhI(OAc) 2 as an oxidant and KHF 2 as an additive, tertiary enamides were converted to isoindolinones via a cascade addition and cyclization process in moderate to good yields.
Abstract
Radical trifluoromethylation and cyclization of tertiary enamides was developed and trifluoromethyl-containing isoindolinones were obtained in moderate to good yields.
