- Record: found
- Abstract: found
- Article: found
Insights into the excited state dynamics of Fe(ii) polypyridyl complexes from variable-temperature ultrafast spectroscopy†
Read this article at
Abstract

Abstract
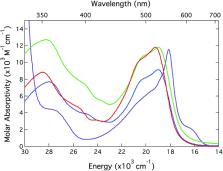
In an effort to better define the nature of the nuclear coordinate associated with excited state dynamics in first-row transition metal-based chromophores, variable-temperature ultrafast time-resolved absorption spectroscopy has been used to determine activation parameters associated with ground state recovery dynamics in a series of low-spin Fe( ii) polypyridyl complexes. Our results establish that high-spin ( 5T 2) to low-spin ( 1A 1) conversion in complexes of the form [Fe(4,4′-di-R-2,2′-bpy′) 3] 2+ (R = H, CH 3, or tert-butyl) is characterized by a small but nevertheless non-zero barrier in the range of 300–350 cm –1 in fluid CH 3CN solution, a value that more than doubles to ∼750 cm –1 for [Fe(terpy) 2] 2+ (terpy = 2,2′:6′,2′′-terpyridine). The data were analyzed in the context of semi-classical Marcus theory. Changes in the ratio of the electronic coupling to reorganization energy (specifically, H ab 4/ λ) reveal an approximately two-fold difference between the [Fe(bpy′) 3] 2+ complexes (∼1/30) and [Fe(terpy) 2] 2+ (∼1/14), suggesting a change in the nature of the nuclear coordinate associated with ground state recovery between these two types of complexes. These experimentally-determined ratios, along with estimates for the 5T 2/ 1A 1 energy gap, yield electronic coupling values between these two states for the [Fe(bpy′) 3] 2+ series and [Fe(terpy) 2] 2+ of 4.3 ± 0.3 cm –1 and 6 ± 1 cm –1, respectively, values that are qualitatively consistent with the second-order nature of high-spin/low-spin coupling in a d 6 ion. In addition to providing useful quantitative information on these prototypical Fe( ii) complexes, these results underscore the utility of variable-temperature spectroscopic measurements for characterizing ultrafast excited state dynamics in this class of compounds.
Related collections
Author and article information
Notes
†Electronic supplementary information (ESI) available. CCDC 1810752 and 1810753. For ESI and crystallographic data in CIF or other electronic format see DOI: 10.1039/c8sc04025g