- Record: found
- Abstract: found
- Article: not found
SERS Based Lateral Flow Immunoassay for Point-of-Care Detection of SARS-CoV-2 in Clinical Samples

Abstract
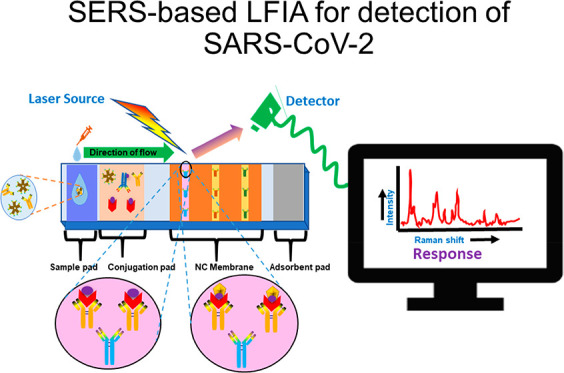
The current scenario, an ongoing pandemic of COVID-19, places a dreadful burden on the healthcare system worldwide. Subsequently, there is a need for a rapid, user-friendly, and inexpensive on-site monitoring system for diagnosis. The early and rapid diagnosis of SARS-CoV-2 plays an important role in combating the outbreak. Although conventional methods such as PCR, RT-PCR, and ELISA, etc., offer a gold-standard solution to manage the pandemic, they cannot be implemented as a point-of-care (POC) testing arrangement. Moreover, surface-enhanced Raman spectroscopy (SERS) having a high enhancement factor provides quantitative results with high specificity, sensitivity, and multiplex detection ability but lacks in POC setup. In contrast, POC devices such as lateral flow immunoassay (LFIA) offer rapid, simple-to-use, cost-effective, reliable platform. However, LFIA has limitations in quantitative and sensitive analyses of SARS-CoV-2 detection. To resolve these concerns, herein we discuss a unique modality that is an integration of SERS with LFIA for quantitative analyses of SARS-CoV-2. The miniaturization ability of SERS-based devices makes them promising in biosensor application and has the potential to make a better alternative of conventional diagnostic methods. This review also demonstrates the commercially available and FDA/ICMR approved LFIA kits for on-site diagnosis of SARS-CoV-2.
Related collections
Most cited references149
- Record: found
- Abstract: found
- Article: not found
A familial cluster of pneumonia associated with the 2019 novel coronavirus indicating person-to-person transmission: a study of a family cluster
- Record: found
- Abstract: found
- Article: not found
SARS-CoV-2 Viral Load in Upper Respiratory Specimens of Infected Patients
- Record: found
- Abstract: found
- Article: not found