- Record: found
- Abstract: found
- Article: found
Efficient Differentiation of Human Pluripotent Stem Cells to Endothelial Progenitors via Small-Molecule Activation of WNT Signaling

Read this article at
Summary
Human pluripotent stem cell (hPSC)-derived endothelial cells and their progenitors may provide the means for vascularization of tissue-engineered constructs and can serve as models to study vascular development and disease. Here, we report a method to efficiently produce endothelial cells from hPSCs via GSK3 inhibition and culture in defined media to direct hPSC differentiation to CD34 +CD31 + endothelial progenitors. Exogenous vascular endothelial growth factor (VEGF) treatment was dispensable, and endothelial progenitor differentiation was β-catenin dependent. Furthermore, by clonal analysis, we showed that CD34 +CD31 +CD117 +TIE-2 + endothelial progenitors were multipotent, capable of differentiating into calponin-expressing smooth muscle cells and CD31 +CD144 +vWF +I-CAM1 + endothelial cells. These endothelial cells were capable of 20 population doublings, formed tube-like structures, imported acetylated low-density lipoprotein, and maintained a dynamic barrier function. This study provides a rapid and efficient method for production of hPSC-derived endothelial progenitors and endothelial cells and identifies WNT/β-catenin signaling as a primary regulator for generating vascular cells from hPSCs.
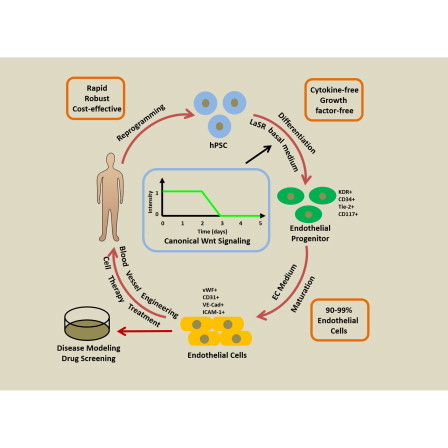
Graphical Abstract
Highlights
-
•
WNT pathway activation directs hPSC differentiation to endothelial progenitors
-
•
hPSC-derived endothelial progenitors can differentiate to endothelial cells
-
•
Purified hPSC-derived endothelial cells are capable of 20 population doublings
-
•
WNT pathway activation permits defined production of endothelial cells from hPSCs
Abstract
In this article, Palecek and colleagues show that GSK3 inhibition in a defined culture medium directs human pluripotent stem cells (hPSCs) to endothelial progenitors. These progenitors further differentiate to cells expressing endothelial markers and exhibiting endothelial phenotypes. This protocol enables rapid and efficient production of hPSC-derived endothelial cells for research and therapeutic applications.
Related collections
Most cited references50
- Record: found
- Abstract: found
- Article: not found
Human induced pluripotent stem cells free of vector and transgene sequences.
- Record: found
- Abstract: found
- Article: not found
Differentiation of embryonic stem cells to clinically relevant populations: lessons from embryonic development.
- Record: found
- Abstract: found
- Article: not found