- Record: found
- Abstract: found
- Article: found
TGF-β signaling in the tumor metabolic microenvironment and targeted therapies
Read this article at
Abstract
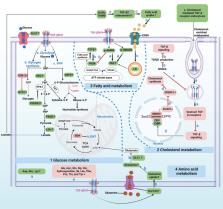
Transforming growth factor-β (TGF-β) signaling has a paradoxical role in cancer progression, and it acts as a tumor suppressor in the early stages but a tumor promoter in the late stages of cancer. Once cancer cells are generated, TGF-β signaling is responsible for the orchestration of the immunosuppressive tumor microenvironment (TME) and supports cancer growth, invasion, metastasis, recurrence, and therapy resistance. These progressive behaviors are driven by an “engine” of the metabolic reprogramming in cancer. Recent studies have revealed that TGF-β signaling regulates cancer metabolic reprogramming and is a metabolic driver in the tumor metabolic microenvironment (TMME). Intriguingly, TGF-β ligands act as an “endocrine” cytokine and influence host metabolism. Therefore, having insight into the role of TGF-β signaling in the TMME is instrumental for acknowledging its wide range of effects and designing new cancer treatment strategies. Herein, we try to illustrate the concise definition of TMME based on the published literature. Then, we review the metabolic reprogramming in the TMME and elaborate on the contribution of TGF-β to metabolic rewiring at the cellular (intracellular), tissular (intercellular), and organismal (cancer-host) levels. Furthermore, we propose three potential applications of targeting TGF-β-dependent mechanism reprogramming, paving the way for TGF-β-related antitumor therapy from the perspective of metabolism.
Related collections
Most cited references316
- Record: found
- Abstract: found
- Article: not found
Understanding the Warburg effect: the metabolic requirements of cell proliferation.
- Record: found
- Abstract: found
- Article: not found