- Record: found
- Abstract: found
- Article: found
Transglutaminase 2 facilitates the distant hematogenous metastasis of breast cancer by modulating interleukin-6 in cancer cells
Read this article at
Abstract
Introduction
Inflammation has been implicated in cancer aggressiveness. As transglutaminase 2 (TG2), which has been associated with inflammatory signaling, has been suggested to play a role in tumor behavior, we propose that TG2 may be an important linker inducing interleukin (IL)-6-mediated cancer-cell aggressiveness, including distant hematogenous metastasis.
Methods
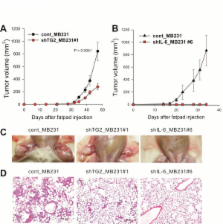
To investigate the role for TG2 and IL-6, TG2-knocked-down and IL-6-knocked-down cancer cells were generated by using shRNA. Human breast cancer cell xenograft model in highly immunocompromised mice and human advanced breast cancer primary tumor tissue microarrays were used in this study.
Results
IL-6 production in human breast cancer cells was dependent on their TG2 expression level. In vitro tumor-sphere formation was dependent on TG2 and downstream IL-6 production from cancer cells. Primary tumor growth in the mammary fat pads and distant hematogenous metastasis into the lung was also dependent on TG2 and downstream IL-6 expression levels. The effect of TG2 expression on human breast cancer distant metastasis was investigated by analyzing a tissue microarray of primary tumors from 412 patients with their clinical data after 7 years. TG2 expression in primary tumor tissue was inversely correlated with recurrence-free survival ( P = 0.019) and distant metastasis-free survival (DMFS) ( P = 0.006) in patients with advanced breast cancer. Furthermore, by using public datasets that included a total of 684 breast cancer patients, we found that the combined high expression of TG2 and IL-6 was associated with shorter DMFS, compared with the high expression of IL-6 only ( P = 0.013).
Related collections
Most cited references31
- Record: found
- Abstract: found
- Article: not found
Epithelial-mesenchymal transitions in development and disease.
- Record: found
- Abstract: found
- Article: not found
IFNgamma and lymphocytes prevent primary tumour development and shape tumour immunogenicity.
- Record: found
- Abstract: found
- Article: not found