- Record: found
- Abstract: found
- Article: found
Clinical and immunological effects of mRNA vaccines in malignant diseases
Read this article at
Abstract
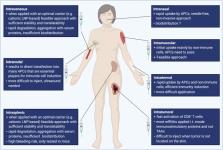
In vitro-transcribed messenger RNA-based therapeutics represent a relatively novel and highly efficient class of drugs. Several recently published studies emphasize the potential efficacy of mRNA vaccines in treating different types of malignant and infectious diseases where conventional vaccine strategies and platforms fail to elicit protective immune responses. mRNA vaccines have lately raised high interest as potent vaccines against SARS-CoV2. Direct application of mRNA or its electroporation into dendritic cells was shown to induce polyclonal CD4+ and CD8+ mediated antigen-specific T cell responses as well as the production of protective antibodies with the ability to eliminate transformed or infected cells. More importantly, the vaccine composition may include two or more mRNAs coding for different proteins or long peptides. This enables the induction of polyclonal immune responses against a broad variety of epitopes within the encoded antigens that are presented on various MHC complexes, thus avoiding the restriction to a certain HLA molecule or possible immune escape due to antigen-loss. The development and design of mRNA therapies was recently boosted by several critical innovations including the development of technologies for the production and delivery of high quality and stable mRNA. Several technical obstacles such as stability, delivery and immunogenicity were addressed in the past and gradually solved in the recent years.
This review will summarize the most recent technological developments and application of mRNA vaccines in clinical trials and discusses the results, challenges and future directions with a special focus on the induced innate and adaptive immune responses.
Related collections
Most cited references204
- Record: found
- Abstract: found
- Article: not found
Cancer immunology. Mutational landscape determines sensitivity to PD-1 blockade in non-small cell lung cancer.
- Record: found
- Abstract: found
- Article: not found
PD-1 blockade induces responses by inhibiting adaptive immune resistance
- Record: found
- Abstract: found
- Article: not found