- Record: found
- Abstract: found
- Article: not found
A Population Pharmacokinetic Model to Predict the Individual Starting Dose of Tacrolimus Following Pediatric Renal Transplantation
Read this article at
Abstract
Background
Multiple clinical, demographic, and genetic factors affect the pharmacokinetics of tacrolimus in children, yet in daily practice, a uniform body-weight based starting dose is used. It can take weeks to reach the target tacrolimus pre-dose concentration.
Objectives
The objectives of this study were to determine the pharmacokinetics of tacrolimus immediately after kidney transplantation and to find relevant parameters for dose individualization using a population pharmacokinetic analysis.
Methods
A total of 722 blood samples were collected from 46 children treated with tacrolimus over the first 6 weeks after renal transplantation. Non-linear mixed-effects modeling (NONMEM ®) was used to develop a population pharmacokinetic model and perform a covariate analysis. Simulations were performed to determine the optimal starting dose and to develop dosing guidelines.
Results
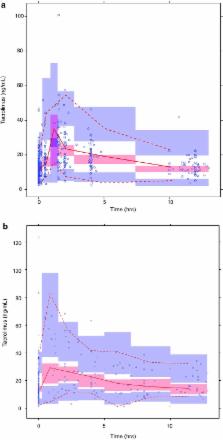
The data were accurately described by a two-compartment model with allometric scaling for bodyweight. Mean tacrolimus apparent clearance was 50.5 L/h, with an inter-patient variability of 25%. Higher bodyweight, lower estimated glomerular filtration rate, and higher hematocrit levels resulted in lower total tacrolimus clearance. Cytochrome P450 3A5 expressers and recipients who received a kidney from a deceased donor had a significantly higher tacrolimus clearance. The model was successfully externally validated. In total, these covariates explained 41% of the variability in clearance. From the significant covariates, the cytochrome P450 3A5 genotype, bodyweight, and donor type were useful to adjust the starting dose to reach the target pre-dose concentration. Dosing guidelines range from 0.27 to 1.33 mg/kg/day.
Related collections
Most cited references47
- Record: found
- Abstract: found
- Article: not found
Prediction-corrected visual predictive checks for diagnosing nonlinear mixed-effects models.
- Record: found
- Abstract: found
- Article: not found
Long-term renal allograft survival in the United States: a critical reappraisal.
- Record: found
- Abstract: found
- Article: not found
