- Record: found
- Abstract: found
- Article: found
Ghrelin transport across the blood–brain barrier can occur independently of the growth hormone secretagogue receptor
Read this article at
Abstract
Objective
The blood–brain barrier (BBB) regulates the entry of substrates and peptides into the brain. Ghrelin is mainly produced in the stomach but exerts its actions in the central nervous system (CNS) by crossing the BBB. Once present in the CNS, ghrelin can act in the hypothalamus to regulate food intake, in the hippocampus to regulate neurogenesis, and in the olfactory bulb to regulate food-seeking behavior. The goal of this study was to determine whether the primary signaling receptor for ghrelin, the growth hormone secretagogue receptor (GHSR), mediates the transport of ghrelin from blood to brain.
Methods
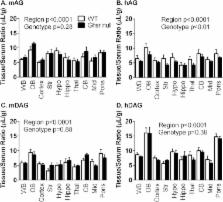
We utilized the sensitive and quantitative multiple-time regression analysis technique to determine the transport rate of mouse and human acyl ghrelin (AG) and desacyl ghrelin (DAG) in wildtype and Ghsr null mice. We also measured the regional distribution of these ghrelin peptides throughout the brain. Lastly, we characterized the transport characteristics of human DAG by measuring the stability in serum and brain, saturability of transport, and the complete transfer across the brain endothelial cell.
Results
We found the transport rate across the BBB of both forms of ghrelin, AG, and DAG, were not affected by the loss of GHSR. We did find differences in the transport rate between the two isoforms, with DAG being faster than AG; this was dependent on the species of ghrelin, human being faster than mouse. Lastly, based on the ubiquitous properties of ghrelin throughout the CNS, we looked at regional distribution of ghrelin uptake and found the highest levels of uptake in the olfactory bulb.
Highlights
Related collections
Most cited references40
- Record: found
- Abstract: found
- Article: not found
Ghrelin octanoylation mediated by an orphan lipid transferase.
- Record: found
- Abstract: found
- Article: not found
Adiponectin, an adipocyte-derived plasma protein, inhibits endothelial NF-kappaB signaling through a cAMP-dependent pathway.
- Record: found
- Abstract: found
- Article: not found