- Record: found
- Abstract: found
- Article: found
Regeneration of Dermis: Scarring and Cells Involved
Read this article at
Abstract
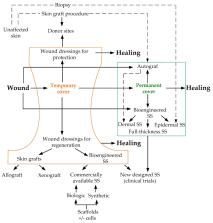
There are many studies on certain skin cell specifications and their contribution to wound healing. In this review, we provide an overview of dermal cell heterogeneity and their participation in skin repair, scar formation, and in the composition of skin substitutes. The papillary, reticular, and hair follicle associated fibroblasts differ not only topographically, but also functionally. Human skin has a number of particular characteristics that are different from murine skin. This should be taken into account in experimental procedures. Dermal cells react differently to skin wounding, remodel the extracellular matrix in their own manner, and convert to myofibroblasts to different extents. Recent studies indicate a special role of papillary fibroblasts in the favorable outcome of wound healing and epithelial-mesenchyme interactions. Neofolliculogenesis can substantially reduce scarring. The role of hair follicle mesenchyme cells in skin repair and possible therapeutic applications is discussed. Participation of dermal cell types in wound healing is described, with the addition of possible mechanisms underlying different outcomes in embryonic and adult tissues in the context of cell population characteristics and extracellular matrix composition and properties. Dermal white adipose tissue involvement in wound healing is also overviewed. Characteristics of myofibroblasts and their activity in scar formation is extensively discussed. Cellular mechanisms of scarring and possible ways for its prevention are highlighted. Data on keloid cells are provided with emphasis on their specific characteristics. We also discuss the contribution of tissue tension to the scar formation as well as the criteria and effectiveness of skin substitutes in skin reconstruction. Special attention is given to the properties of skin substitutes in terms of cell composition and the ability to prevent scarring.
Related collections
Most cited references204
- Record: found
- Abstract: found
- Article: not found
Effects of substrate stiffness on cell morphology, cytoskeletal structure, and adhesion.
- Record: found
- Abstract: found
- Article: not found