- Record: found
- Abstract: found
- Article: found
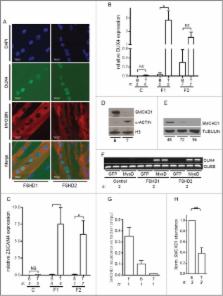
Increased DUX4 expression during muscle differentiation correlates with decreased SMCHD1 protein levels at D4Z4
Read this article at
Abstract
Facioscapulohumeral muscular dystrophy is caused by incomplete epigenetic repression of the transcription factor DUX4 in skeletal muscle. A copy of DUX4 is located within each unit of the D4Z4 macrosatellite repeat array and its derepression in somatic cells is caused by either repeat array contraction (FSHD1) or by mutations in the chromatin repressor SMCHD1 (FSHD2). While DUX4 expression has thus far only been detected in FSHD muscle and muscle cell cultures, and increases with in vitro myogenic differentiation, the D4Z4 chromatin structure has only been studied in proliferating myoblasts or non-myogenic cells. We here show that SMCHD1 protein levels at D4Z4 decline during muscle cell differentiation and correlate with DUX4 derepression. In FSHD2, but not FSHD1, the loss of SMCHD1 repressor activity is partially compensated by increased Polycomb Repressive Complex 2 (PRC2)–mediated H3K27 trimethylation at D4Z4, a situation that can be mimicked by SMCHD1 knockdown in control myotubes. In contrast, moderate overexpression of SMCHD1 results in DUX4 silencing in FSHD1 and FSHD2 myotubes demonstrating that DUX4 derepression in FSHD is reversible. Together, we show that in FSHD1 and FSHD2 the decline in SMCHD1 protein levels during muscle cell differentiation renders skeletal muscle sensitive to DUX4.
Related collections
Most cited references37
- Record: found
- Abstract: found
- Article: not found
A unifying genetic model for facioscapulohumeral muscular dystrophy.
- Record: found
- Abstract: found
- Article: not found
Digenic inheritance of an SMCHD1 mutation and an FSHD-permissive D4Z4 allele causes facioscapulohumeral muscular dystrophy type 2
- Record: found
- Abstract: found
- Article: not found