- Record: found
- Abstract: found
- Article: found
Mechanism of action of a T cell-dependent bispecific antibody as a breakthrough immunotherapy against refractory colorectal cancer with an oncogenic mutation
Read this article at
Abstract
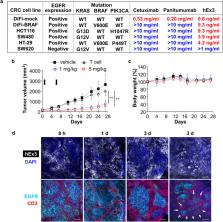
T cell-dependent bispecific antibody (TDB)-induced T cell activation, which can eliminate tumor cells independent of MHC engagement, is expected to be a novel breakthrough immunotherapy against refractory cancer. However, the mechanism of action of TDBs has not been fully elucidated thus far. We focused on TDB-induced T cell–tumor cell contact as an important initial step in direct T cell-mediated tumor cell killing via transport of cytotoxic cell proteases (e.g., granzymes) with or without immunological synapse formation. Using an anti-EGFR/CD3 TDB, hEx3, we visualized and quantified T cell–tumor cell contact and demonstrated a correlation between the degree of cell contact and TDB efficacy. We also found that cytokines, including interferon-gamma (IFNγ) and tumor necrosis factor-alpha (TNFα) secreted by activated T cells, damaged tumor cells in a cell contact-independent manner. Moreover, therapeutic experiences clearly indicated that hEx3, unlike conventional anti-EGFR antibodies, was effective against colorectal cancer (CRC) cells with mutant KRAS, BRAF, or PIK3CA. In a pharmacokinetic analysis, T cells spread gradually in accordance with the hEx3 distribution within tumor tissue. Accordingly, we propose that TDBs should have four action steps: 1st, passive targeting via size-dependent tumor accumulation; 2nd, active targeting via specific binding to tumor cells; 3rd, T cell redirection toward tumor cells; and 4th, TDB-induced cell contact-dependent (direct) or -independent (indirect) tumor cell killing. Finally, our TDB hEx3 may be a promising reagent against refractory CRC with an oncogenic mutation associated with a poor prognosis.
Related collections
Most cited references36
- Record: found
- Abstract: found
- Article: not found
Cancer statistics, 2018
- Record: found
- Abstract: found
- Article: not found
A new concept for macromolecular therapeutics in cancer chemotherapy: mechanism of tumoritropic accumulation of proteins and the antitumor agent smancs.
- Record: found
- Abstract: found
- Article: not found