- Record: found
- Abstract: found
- Article: not found
Mesoscale All-Atom Influenza Virus Simulations Suggest New Substrate Binding Mechanism
Read this article at
Abstract
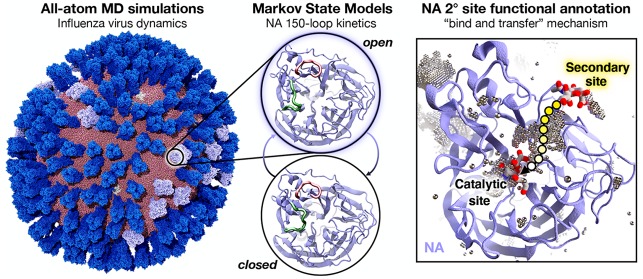
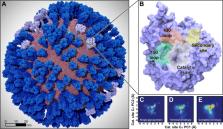
Influenza virus circulates in human, avian, and swine hosts, causing seasonal epidemic and occasional pandemic outbreaks. Influenza neuraminidase, a viral surface glycoprotein, has two sialic acid binding sites. The catalytic (primary) site, which also binds inhibitors such as oseltamivir carboxylate, is responsible for cleaving the sialic acid linkages that bind viral progeny to the host cell. In contrast, the functional annotation of the secondary site remains unclear. Here, we better characterize these two sites through the development of an all-atom, explicitly solvated, and experimentally based integrative model of the pandemic influenza A H1N1 2009 viral envelope, containing ∼160 million atoms and spanning ∼115 nm in diameter. Molecular dynamics simulations of this crowded subcellular environment, coupled with Markov state model theory, provide a novel framework for studying realistic molecular systems at the mesoscale and allow us to quantify the kinetics of the neuraminidase 150-loop transition between the open and closed states. An analysis of chloride ion occupancy along the neuraminidase surface implies a potential new role for the neuraminidase secondary site, wherein the terminal sialic acid residues of the linkages may bind before transfer to the primary site where enzymatic cleavage occurs. Altogether, our work breaks new ground for molecular simulation in terms of size, complexity, and methodological analyses of the components. It also provides fundamental insights into the understanding of substrate recognition processes for this vital influenza drug target, suggesting a new strategy for the development of anti-influenza therapeutics.
Abstract
Molecular dynamics simulations and chloride ion analyses provide fundamental insights into the understanding of substrate recognition processes for two sialic binding sites of influenza neuraminidase.
Related collections
Most cited references51

- Record: found
- Abstract: found
- Article: found
Influenza Hemagglutinin and Neuraminidase Membrane Glycoproteins*
- Record: found
- Abstract: found
- Article: not found
Everything you wanted to know about Markov State Models but were afraid to ask.
- Record: found
- Abstract: found
- Article: not found
