- Record: found
- Abstract: found
- Article: found
Non-small cell lung cancer cells survived ionizing radiation treatment display cancer stem cell and epithelial-mesenchymal transition phenotypes
Read this article at
Abstract
Ionizing radiation (IR) is used for patients diagnosed with unresectable non small cell lung cancer (NSCLC), however radiotherapy remains largely palliative due to radioresistance. Cancer stem cells (CSCs), as well as epithelial-mesenchymal transition (EMT), may contribute to drug and radiation resistance mechanisms in solid tumors. Here we investigated the molecular phenotype of A549 and H460 NSCLC cells that survived treatment with IR (5Gy) and are growing as floating tumor spheres and cells that are maintained in a monolayer after irradiation.
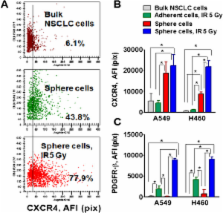
Non-irradiated and irradiated cells were collected after one week, seeded onto ultra low attachment plates and propagated as tumor spheres. Bulk NSCLC cells which survived radiation and grew in spheres express cancer stem cell surface and embryonic stem cell markers and are able to self-renew, and generate differentiated progeny. These cells also have a mesenchymal phenotype. Particularly, the radiation survived sphere cells express significantly higher levels of CSC markers (CD24 and CD44), nuclear β-catenin and EMT markers (Snail1, Vimentin, and N-cadherin) than non-irradiated lung tumor sphere cells. Upregulated levels of Oct-4, Sox2 and beta-catenin were detected in H460 cells maintained in a monolayer after irradiation, but not in radiation survived adherent A459 cells.
PDGFR-beta was upregulated in radiation survived sphere cells and in radiation survived adherent cells in both A549 and H460 cell lines. Combining IR treatment with axitinib or dasatinib, inhibitors with anti-PDFGR activity, potentiates the efficacy of NSCLC radiotherapy in vitro.
Our findings suggest that radiation survived cells have a complex phenotype combining the properties of CSCs and EMT. CD44, SNAIL and PDGFR-beta are dramatically upregulated in radiation survived cells and might be considered as markers of radiotherapy response in NSCLC.
Related collections
Most cited references47
- Record: found
- Abstract: found
- Article: not found
Highly tumorigenic lung cancer CD133+ cells display stem-like features and are spared by cisplatin treatment.
- Record: found
- Abstract: found
- Article: not found
Aldehyde dehydrogenase 1 is a tumor stem cell-associated marker in lung cancer.
- Record: found
- Abstract: found
- Article: not found