- Record: found
- Abstract: found
- Article: found
What’s old is new: Valacyclovir for the treatment of pityriasis rosea, a retrospective case series
case-report
Gina M. Ashforth , MD
a
,
∗ ,
Sara Youssef , MPH, MBS
b ,
Veena Bhagavathi , BA
b ,
Cindy Wassef , MD
a ,
Jason H. Miller , MD
a
,
c
20 May 2023
Read this article at
There is no author summary for this article yet. Authors can add summaries to their articles on ScienceOpen to make them more accessible to a non-specialist audience.
Abstract
Introduction
Pityriasis rosea (PR) is a common and self-limiting papulosquamous skin condition.
It presents with the most distinguishable sign of a larger “herald” patch with a collarette
of scale at the margins, followed by smaller, finer, bilateral erythematous scaly
plaques. The lesions commonly occur over the trunk and extremities in a blaschkolinear
“Christmas tree” distribution. PR is most encountered as a springtime eruption and
typically affects children and adults (age, 10-35 years), with a peak in adolescence.
The estimated prevalence of PR in the United States was found to be 0.3% to 3% at
dermatologic centers and evenly distributed among both sexes as 0.13% in females and
0.14% in males.
1
Upon diagnosis of PR, patients may be informed that the condition is self-limiting,
noncontagious, and unlikely to recur. A typical course develops and resolves on average
within 6 to 8 weeks without therapy.2, 3, 4 However, the cutaneous eruption has been
reported to last for 3 to 6 months.
1
,
5
,
6
For some patients, the “watchful waiting” approach may not be sufficient due to symptomology
and psychosocial distress associated with the appearance of lesions. Symptomatic treatments
for symptoms such as pruritus include topical corticosteroids, oral antihistamines,
and anti-itch lotions. Alternative potential treatments include UV-B phototherapy;
antivirals, such as acyclovir; and macrolide antibiotics,
3
,
4
,
7
with only modest success in clearing the eruption. Of the macrolides, a triple-blinded
study on erythromycin 250 mg 4 times daily for 2 weeks resulted in remission after
2 weeks in 65% of patients. Gastrointestinal upset was observed in 10% of patients.
4
A bilateral comparison study of UV-B in patients with PR notes clearance on the treated
half of the body after an average of 4.7 to 6.8 treatments, with notable adverse effect
in some patients of slight tenderness and dryness of the skin.
7
Seasonal variations and clustering in close communities suggest an infectious agent
as the inciting factor for PR. Although human herpesviruses (HHVs) (HHV-6 and HHV-7)
have often been implicated in a causal relationship with PR,
8
the exact cause remains unknown. Additionally, PR-like eruptions have been reported
with COVID-19,
9
vaccinations (ie, bacille Calmette-Guerin vaccine, influenza, diphtheria, smallpox,
hepatitis B, and COVID-19), and medications (ie, gold, barbiturates, captopril, and
clonidine).
10
There are case reports of successful treatment of PR with acyclovir based on the theory
of the pathogenic involvement of HHV-6 and HHV-7, particularly when administered within
the first week of symptom onset.
11
There are additional but fewer reports of using valacyclovir (known pharmaceutically
as Valacyclovir), which is the prodrug of acyclovir.
12
The efficacy of valacyclovir as treatment for PR has so far been reported in 1 case
series of 3 patients: while watchful waiting led to worsening symptoms, treatment
with valacyclovir 1 g 3 times daily for 1 week led to resolution of the condition
within 2 to 3 weeks.
13
The authors have proposed valacyclovir as an alternative therapeutic option due to
its less frequent dosing and a more favorable safety profile when compared with those
of acyclovir. The dosing schedule of valacyclovir used in this series was adapted
from the established treatment regimen for herpes zoster infection (shingles), which
consists of oral valacyclovir 1 g 3 times daily for 7 days, which is the standard
dose without renal adjustment.
Materials and methods
A retrospective chart review was conducted on 9 patient cases from 2 clinical dermatology
sites, 1 academic center and 1 private practice, both in New Jersey. The patients
are adult patients aged 22 to 72 years who were evaluated between the years 2018 and
2022. The patients were then seen on follow-up weeks to months after treatment, and
improvement in the appearance of rash and symptoms were documented along with any
medication adverse events or intolerance. Clinical photographs were taken to monitor
the progress.
Response to treatment was characterized in 3 tiers: (1) “none” was defined as lack
of response or persistence of lesions and symptoms after full treatment course, (2)
“significant improvement” was defined as improvement in the appearance and symptomology
of primary lesions but a lack of complete clearance, and (3) “resolved” was defined
as clearance of primary lesions with absence of residual symptoms and with either
absence or presence of mild postinflammatory secondary changes.
Case presentation
For a summary of cases, please refer to Table I.
Table I
Clinical characteristics of patients with pityriasis rosea who were treated with oral
valacyclovir
Age (y)
Sex
Diagnosis
Time of symptom onset prior to initial presentation
Timing of follow-up
Status of PR eruption
Side effects from valacyclovir
Confounding treatments
28
F
Clinical PR
2 wk
3 wk
Resolved
None
None
22
F
Clinical PR
3 d
6 wk
Resolved
None
Triamcinolone 0.1% cream
32
F
Clinical PR
2 wk
10 d
Resolved
None
None
24
M
Clinical inverse PR
2 wk
2 wk
Significant improvement
None
None
72
M
Clinical PR
4 wk
4 wk
Resolved
None
None
27
M
Clinical PR
3 d
2 wk
Significant improvement
None
Triamcinolone 0.1% cream
67
F
Clinical PR
1 wk
2 wk
Resolved
None
Triamcinolone 0.1% cream
35
F
Clinical PR
2 wk
2 wk
Significant improvement
None
Triamcinolone 0.1% cream
29
F
Biopsy-proven PR
12 wk
2 wk
Significant improvement
None
None; flare was thought to be secondary to COVID-19 vaccine
PR, Pityriasis rosea.
Case 1
A 28-year-old woman presented with a 2-week history of a rash diagnosed clinically
as PR. She was started on valacyclovir 1 g 3 times daily by mouth for 7 days. She
was prescribed no other topical or oral treatments. On follow-up 3 weeks after the
initial presentation, her rash was resolved. She reported no side effects from valacyclovir.
The total time from onset to follow-up after treatment was 5 weeks.
Case 2
A 22-year-old woman presented with a 3-day history of a rash diagnosed clinically
as PR. She was started on valacyclovir 1 g 3 times daily by mouth for 7 days. She
was also prescribed topical triamcinolone 0.1% cream. On follow-up 6 weeks after the
initial presentation, her rash was resolved. She reported no side effects from valacyclovir.
The total time from onset to follow-up after treatment was 6 weeks and 3 days.
Case 3
A 32-year-old woman presented with a 2-week history of a rash diagnosed clinically
as PR. She was started on valacyclovir 1 g 3 times daily by mouth for 7 days. She
was prescribed no other topical or oral treatments. On follow-up 10 days after the
initial presentation, her rash was resolved. She reported no side effects from valacyclovir.
The total time from onset to follow-up after treatment was 3 weeks and 3 days.
Case 4
A 24-year-old man presented with a 2-week history of a rash diagnosed clinically as
PR. He was started on valacyclovir 1 g 3 times daily by mouth for 7 days. He was prescribed
no other topical or oral treatments. On follow-up 2 weeks after the initial presentation,
his rash was significantly improved. He reported no side effects from valacyclovir.
The total time from onset to follow-up after treatment was 4 weeks.
Case 5
A 72-year-old man presented with a 4-week history of a rash diagnosed clinically as
PR. He was started on valacyclovir 1 g 3 times daily by mouth for 7 days. He was prescribed
no other topical or oral treatments. On follow-up 4 weeks after the initial presentation,
his rash was resolved. He reported no side effects from valacyclovir. The total time
from onset to follow-up after treatment was 8 weeks.
Case 6
A 27-year-old man presented with a 3-day history of a rash diagnosed clinically as
PR. He was started on valacyclovir 1 g 3 times daily by mouth for 7 days. He was also
prescribed topical triamcinolone 0.1% cream. On follow-up 2 weeks after the initial
presentation, his rash was significantly improved. He reported no side effects from
valacyclovir. The total time from onset to follow-up after treatment was 2 weeks and
3 days.
Case 7
A 67-year-old woman presented with a 1-week history of a rash diagnosed clinically
as PR. She was started on valacyclovir 1 g 3 times daily by mouth for 7 days. She
was also prescribed topical triamcinolone 0.1% cream. On follow-up 2 weeks after the
initial presentation, her rash was resolved. She reported no side effects from valacyclovir.
The total time from onset to follow-up after treatment was 3 weeks.
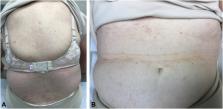
Please refer to Figs 1 and 2 for before-and-after clinical photographs.
Fig 1
Initial presentation of patient 7 with a herald patch and round erythematous thin
scaly plaques on the (A) posterior aspect of the trunk and (B) anterior aspect of
the trunk.
Fig 2
Patient 7 2 weeks after initiating valacyclovir therapy (3 weeks total time elapsed
after rash onset) with resolution and only mild postinflammatory changes present on
the (A) posterior aspect of the trunk and (B) anterior aspect of the trunk.
Case 8
A 35-year-old woman presented with a 2-week history of a rash diagnosed clinically
as PR. She was started on valacyclovir 1 g 3 times daily by mouth for 7 days. She
was also prescribed topical triamcinolone 0.1% cream. On follow-up 2 weeks after the
initial presentation, her rash was significantly improved. She reported no side effects
from valacyclovir. The total time from onset to follow-up after treatment was 4 weeks.
Case 9
A 29-year-old woman presented with a 12-week history of a rash that started days after
her COVID-19 booster vaccination. Diagnosis of PR was made clinically and with histopathologic
evaluation. She was started on valacyclovir 1 g 3 times daily by mouth for 7 days.
She was prescribed no other topical or oral treatments. On follow-up 2 weeks after
the initial presentation, her rash was greatly improved. She reported no side effects
from valacyclovir. The total time from onset to follow-up after treatment was 14 weeks.
Discussion
PR is a common, acute, and relatively benign papulosquamous dermatosis that typically
self-resolves in 6 to 8 weeks without intervention. Prolonged duration of this rash
has been reported to be up to 6 months. Due to the self-limiting nature of the condition,
there is no standard of care for the active treatment of PR. Common therapeutics,
such as topical steroids and oral antihistamines, are targeted to symptomology if
present. Nonetheless, the watchful waiting approach may not be acceptable to some
patients due to the psychosocial implications of the appearance of the rash, which
may be confused with an infectious dermatosis, such as a dermatophytid, by the layperson.
As with many dermatologic conditions, the patient’s quality of life must be considered,
and some cases may necessitate active intervention.
4
The proposed link between HHV-6/HHV-7 and PR has led to the suggested use of antivirals
for treatment.
14
The use of acyclovir for treatment of PR began appearing in the literature shortly
after the HHV association was established, with mixed data in efficacy. In the authors’
opinion, the use of valacyclovir poses certain significant advantages over the use
of acyclovir. Unlike acyclovir, which is dosed 3 to 5 times daily depending on indication,
valacyclovir is offered orally in 500- and 1000-mg tablets and is typically taken
1 to 3 times daily. Valacyclovir is a generic medication associated with a relatively
low out-of-pocket cost to patients, as listed on a coupon-based website with a cost
as low as $6.76 for 21 1-g tablets. In the authors’ experience, it has been covered
by various insurance companies for this rash without requirement of prior authorization.
It is noted that valacyclovir is the prodrug of acyclovir resulting in similar safety
profiles. Some common side effects of valacyclovir include headache, dizziness, nausea,
vomiting, joint pain, and rash (valacyclovir). Severe rash, such as Stevens-Johnson
syndrome–toxic epidermal necrolysis spectrum, has been described in 1 case report
after treatment with acyclovir, but, to our knowledge, there are no reports to date
of this severe cutaneous adverse reaction after valacyclovir use.
15
No patient in our case series reported adverse effects or side effects from taking
valacyclovir for the specified dose and duration. No patient had to discontinue the
week-long course due to medication side effects. No patient required renal dosing
adjustment.
All patients in this case series reported either significant improvement (44.4%) or
total resolution (55.6%) with the proposed treatment, suggesting that valacyclovir
may be considered a reasonable treatment option for PR. Six of 9 patients treated
with valacyclovir had complete resolution or significant improvement of their rash
in a duration of <6 weeks, which is shorter than the cited duration of 6 weeks to
6 months with spontaneous resolution. Of 3 patients who were monitored with a rash
for longer than 6 weeks, patient 9 suffered with the eruption for 12 weeks before
evaluation in our clinic. Despite flaring after COVID-19 vaccination and failing potent
topical corticosteroid treatment, her rash and quality of life were greatly improved
at her 2-week follow-up.
Conclusion
Limitations to this case series include lack of matched control or comparison group,
small sample size, and lack of histopathologic confirmation of diagnosis for all patients.
The authors do note that there remains uncertainty as to whether the eruption cleared
spontaneously or from the valacyclovir. It is believed that clearance of the PR eruption
in 6 of our 9 cases in <6 weeks is a promising metric, especially when considering
the safe and cost-effective nature of a short course of oral valacyclovir. Future
work that would ideally include randomized control trials to assess the comparable
effectiveness among valacyclovir, acyclovir, and placebo for PR as well as studies
with larger sample sizes is needed.
Conflicts of interest
None disclosed.
Related collections
Most cited references15
- Record: found
- Abstract: found
- Article: found
Pityriasis rosea as a cutaneous manifestation of COVID‐19 infection
Amir Ehsani, Maryam Nasimi, Zeinab Bigdelo (2020)
- Record: found
- Abstract: found
- Article: not found
Pityriasis rosea is associated with systemic active infection with both human herpesvirus-7 and human herpesvirus-6.
Sharon E. Jacob, Takahiro Watanabe, B Black … (2002)
- Record: found
- Abstract: found
- Article: found
Pityriasis Rosea: A Comprehensive Classification
Francesco Drago, Giulia Ciccarese, Alfredo Rebora … (2016)