- Record: found
- Abstract: found
- Article: found
miR-22 Is a Novel Mediator of Vascular Smooth Muscle Cell Phenotypic Modulation and Neointima Formation
Read this article at
Abstract
Supplemental Digital Content is available in the text.
Abstract
Background:
MicroRNA-22 (miR-22) has recently been reported to play a regulatory role during vascular smooth muscle cell (VSMC) differentiation from stem cells, but little is known about its target genes and related pathways in mature VSMC phenotypic modulation or its clinical implication in neointima formation following vascular injury.
Methods:
We applied a wire-injury mouse model, and local delivery of AgomiR-22 or miR-22 inhibitor, as well, to explore the therapeutic potential of miR-22 in vascular diseases. Furthermore, normal and diseased human femoral arteries were harvested, and various in vivo, ex vivo, and in vitro models of VSMC phenotype switching were conducted to examine miR-22 expression during VSMC phenotype switching.
Results:
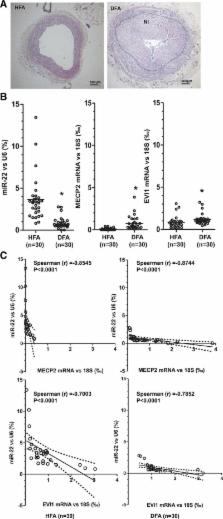
Expression of miR-22 was closely regulated during VSMC phenotypic modulation. miR-22 overexpression significantly increased expression of VSMC marker genes and inhibited VSMC proliferation and migration, whereas the opposite effect was observed when endogenous miR-22 was knocked down. As expected, 2 previously reported miR-22 target genes, MECP2 (methyl-CpG binding protein 2) and histone deacetylase 4, exhibited a regulatory role in VSMC phenotypic modulation. A transcriptional regulator and oncoprotein, EVI1 (ecotropic virus integration site 1 protein homolog), has been identified as a novel miR-22 target gene in VSMC phenotypic modulation. It is noteworthy that overexpression of miR-22 in the injured vessels significantly reduced the expression of its target genes, decreased VSMC proliferation, and inhibited neointima formation in wire-injured femoral arteries, whereas the opposite effect was observed with local application of a miR-22 inhibitor to injured arteries. We next examined the clinical relevance of miR-22 expression and its target genes in human femoral arteries. We found that miR-22 expression was significantly reduced, whereas MECP2 and EVI1 expression levels were dramatically increased, in diseased in comparison with healthy femoral human arteries. This inverse relationship between miR-22 and MECP2 and EVI1 was evident in both healthy and diseased human femoral arteries.
Related collections
Most cited references51
- Record: found
- Abstract: found
- Article: not found
miR-145 and miR-143 Regulate Smooth Muscle Cell Fate Decisions
- Record: found
- Abstract: found
- Article: not found
Smooth muscle cell phenotypic switching in atherosclerosis.
- Record: found
- Abstract: found
- Article: not found