- Record: found
- Abstract: found
- Article: found
Prospects and challenges of extracellular vesicle-based drug delivery system: considering cell source
Read this article at
Abstract
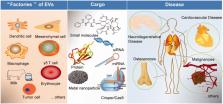
Extracellular vesicles (EVs), including exosomes, microvesicles, and apoptotic bodies, are nanosized membrane vesicles derived from most cell types. Carrying diverse biomolecules from their parent cells, EVs are important mediators of intercellular communication and thus play significant roles in physiological and pathological processes. Owing to their natural biogenesis process, EVs are generated with high biocompatibility, enhanced stability, and limited immunogenicity, which provide multiple advantages as drug delivery systems (DDSs) over traditional synthetic delivery vehicles. EVs have been reported to be used for the delivery of siRNAs, miRNAs, protein, small molecule drugs, nanoparticles, and CRISPR/Cas9 in the treatment of various diseases. As a natural drug delivery vectors, EVs can penetrate into the tissues and be bioengineered to enhance the targetability. Although EVs’ characteristics make them ideal for drug delivery, EV-based drug delivery remains challenging, due to lack of standardized isolation and purification methods, limited drug loading efficiency, and insufficient clinical grade production. In this review, we summarized the current knowledge on the application of EVs as DDS from the perspective of different cell origin and weighted the advantages and bottlenecks of EV-based DDS.
Related collections
Most cited references112
- Record: found
- Abstract: found
- Article: not found
Mesenchymal stem cells derived from dental tissues vs. those from other sources: their biology and role in regenerative medicine.
- Record: found
- Abstract: found
- Article: not found
Decisions about dendritic cells: past, present, and future.
Author and article information
Comments
Comment on this article
 Smart Citations
Smart CitationsSee how this article has been cited at scite.ai
scite shows how a scientific paper has been cited by providing the context of the citation, a classification describing whether it supports, mentions, or contrasts the cited claim, and a label indicating in which section the citation was made.