- Record: found
- Abstract: found
- Article: found
Biomarkers in Hepatocellular Carcinoma: Diagnosis, Prognosis and Treatment Response Assessment
Read this article at
Abstract
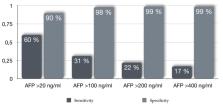
Hepatocellular carcinoma (HCC) is one of the main cancer-related causes of death worldwide. Thus, there is a constant search for improvement in screening, diagnosis, and treatment strategies to improve the prognosis of this malignancy. The identification of useful biomarkers for surveillance and early HCC diagnosis is still deficient, with available serum biomarkers showing low sensitivity and heterogeneous specificity despite different cut-off points, even when assessed longitudinally, or with a combination of serum biomarkers. In contrast, HCC biomarkers used for prognostic (when associated with clinical outcomes) or predictive purposes (when associated with treatment response) may have an increased clinical role in the near future. Furthermore, some serum biomarkers are already implicated as a treatment selection tool, whether to provide access to certain therapies or to assess clinical benefit after treatment. In the present review we will discuss the clinical utility and foreseen future of HCC biomarkers implicated in surveillance, diagnosis, prognosis, and post-treatment assessment.
Related collections
Most cited references139
- Record: found
- Abstract: found
- Article: found
Comprehensive and Integrative Genomic Characterization of Hepatocellular Carcinoma
- Record: found
- Abstract: found
- Article: not found
Surveillance Imaging and Alpha Fetoprotein for Early Detection of Hepatocellular Carcinoma in Patients With Cirrhosis: A Meta-analysis
- Record: found
- Abstract: found
- Article: not found