- Record: found
- Abstract: found
- Article: not found
Profiling B cell immunodominance after SARS-CoV-2 infection reveals antibody evolution to non-neutralizing viral targets
Read this article at
Abstract
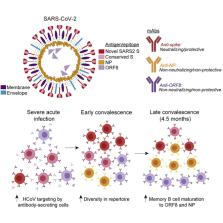
Dissecting the evolution of memory B cells (MBCs) against SARS-CoV-2 is critical for understanding antibody recall upon secondary exposure. Here, we utilized single-cell sequencing to profile SARS-CoV-2-reactive B cells in 38 COVID-19 patients. Using oligo-tagged antigen baits, we isolated B cells specific to the SARS-CoV-2 spike, nucleoprotein (NP), open reading frame 8 (ORF8), and endemic coronavirus (HCoV) spike proteins. SARS-CoV-2 spike-specific cells were enriched in the memory compartment of acutely infected and convalescent patients several months post-symptom onset. With severe acute infection, substantial populations of endemic HCoV-reactive antibody-secreting cells were identified and possessed highly mutated variable genes, signifying preexisting immunity. Finally, MBCs exhibited pronounced maturation to NP and ORF8 over time, especially in older patients. Monoclonal antibodies against these targets were non-neutralizing and non-protective in vivo. These findings reveal antibody adaptation to non-neutralizing intracellular antigens during infection, emphasizing the importance of vaccination for inducing neutralizing spike-specific MBCs.
Graphical Abstract
Abstract
Dugan et al. utilize a multi-antigen bait sorting and single cell sequencing approach to profile B cell immunodominance upon SARS-CoV-2 infection, revealing a dynamic response that evolves toward internal virus proteins over time. Antibodies to internal proteins were non-protective in vivo, suggesting vaccination may generate superior anti-spike immunological memory.

