- Record: found
- Abstract: found
- Article: found
One-step generation of knockout pigs by zygote injection of CRISPR/Cas system
letter
Read this article at
There is no author summary for this article yet. Authors can add summaries to their articles on ScienceOpen to make them more accessible to a non-specialist audience.
Abstract
Dear Editor,
The pig is an important livestock for food supply and an ideal model for various human
diseases. Efficient and precise genetic engineering in pigs holds great promise in
agriculture and biomedicine
1
. Using currently available approach, generating specific gene modifications in pigs
requires two steps. First, site-specific nucleases such as zinc finger nucleases (ZFNs)
and transcription activator-like effector nucleases (TALENs) are used to generate
targeted mutations in pig somatic cells. Then the engineered somatic nucleus is used
to generate cloned animals using somatic cell nuclear transfer (SCNT) technology
2,3
. The complex design and generation of ZFNs and TALENs, as well as the technical challenges
of SCNT, greatly limit the application of this method.
Recently a new type of site-specific nuclease was identified in CRISPR (clustered
regularly interspaced short palindromic repeat)/Cas (CRISPR-associated) RNA-based
adaptive immune system in bacteria and archaea
4
. The Cas9 endonuclease from Streptococcus pyogenes type II CRISPR/Cas system can
be engineered to produce targeted genome modification under the guidance of a synthetic
single guide RNA (sgRNA) with simple base-pair complementarities with a target genomic
DNA sequence
5,6,7
. Although highly efficient one-step generation of mice and rats carrying single or
multiple mutations have been achieved through direct injection of CRISPR/Cas system
into one-cell embryo
8,9
, it remains to be tested whether the same strategy could be applied to generation
of genome-engineered large animals. Here we report the efficient generation of biallelic
knockout pigs in one step by direct cytoplasmic injection of Cas9 mRNA and sgRNA into
zygotes.
To demonstrate the application of genome engineering in disease modeling and agriculture,
we chose to target vWF gene, whose deficiency in human causes severe von Willebrand
disease (vWD)
10
(Supplementary information, Figure S1). As mouse model does not fully recapitulate
the disease phenotype and response to treatment in human patients, a large animal
model with similar size of organ and circulatory system is highly desirable. In food
industry, bleeding is an important procedure before meat production, as the leftover
blood is an ideal medium for bacterial growth. vWF knockout could improve bleeding
efficiency for slaughtering procedures and blood collection.
We designed an sgRNA targeting exon 5 of the pig vWF gene, which lies in the first
trysin-inhibitor-like domain and its mutation could lead to the loss of function of
vWF protein. The in vitro developmental efficiencies of embryos injected with Cas9
mRNA/sgRNA (∼79%) and embryos injected with water (∼77%) were both very high and comparable
with each other, suggesting that the microinjection manipulation and the Cas9 mRNA/sgRNA
had little effect on pig early embryonic development (Supplementary information, Table
S1).
We next transferred the injected embryos into surrogate pigs to produce piglets. A
total of 76 injected embryos were transplanted into 5 surrogate mothers in 5 independent
experiments. Three surrogates were successfully pregnant and delivered a total of
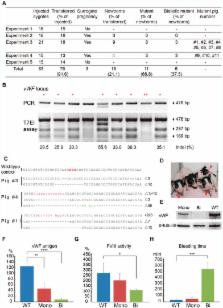
16 piglets (Figure 1A). T7 endonuclease I (T7EI) assay showed that 10 piglets contained
insertions or deletions (indels) in the targeting site, indicating that vWF mutation
efficiency reached about 63% (10/16) (Figure 1A and 1B). We further analyzed the genotype
by Sanger sequencing of the PCR products containing the targeting site of each individual
piglet. The sequencing results confirmed the indels and revealed that one mutant pig
yielding uncut PCR product in the T7EI assay (fourth lane in Figure 1B) contained
only one type of mutant allele. Most pigs contained more than two different alleles
(Figure 1C, pig #4 and #11), suggesting that the Cas9-mediated DNA cleavage may occur
at embryonic stage later than the one-cell stage, as previously observed with ZFN,
TALEN and CRISPR-mediated targeting
11
. In each pig, if no wild-type allele was detected by sequencing, we categorized the
pig as biallelic mutant. If both wild-type and mutant alleles were observed in the
sequence reads, we categorized the pig as monoallelic mutant, although the ratio of
wild-type to mutant alleles might not always be 1. In total, we identified six vWF
biallelic mutant piglets and five monoallelic mutant piglets (11/16, 68%) (Figure
1A and 1C). Of these piglets, two died shortly after birth and the other 14 survived
until now (Figure 1D). The high birth rate (16/76, 21%) and survival rate (14/16,
88%) indicated that Cas9 mRNA/sgRNA had little toxicity to pig embryonic development
(Figure 1A). These results demonstrate that the CRISPR/Cas system could efficiently
generate both monoallelic and biallelic mutant pigs in one step through direct zygote
injection.
We further examined whether the vWF mutations cause the vWD phenotypes. Western blot
analysis showed that the vWF expression in lung was largely reduced in monoallelic
mutant pigs, and was undetectable in biallelic mutant pigs, confirming the vWF knockout
at protein levels (Figure 1E). We next performed hematological analysis with pigs
having different genotypes. The plasma vWF antigen level was significantly reduced
in the monoallelic mutant pig group, and was nearly undetectable in biallelic mutant
group (Figure 1F). In human vWD, the level of coagulation factor FVIII may be reduced
due to rapid degradation in the absence of vWF
10
. Consistently, the activated partial thromboplastin time (APTT)-based assay for FVIII
activity showed a significantly lower activity in the biallelic mutant group, indicating
that FVIII level was decreased in the plasma of vWF-null pigs (Figure 1G). No significant
differences were observed between wild-type and knockout pigs with regard to other
parameters in the hematological analysis (Supplementary information, Table S2). The
most prominent phenotype resulting from vWF mutation is a severe bleeding tendency
10
. We found that the bleeding time of biallelic mutant pigs (512 ± 121 min) was much
longer than that of wild-type pigs (34 ± 11 min) (Figure 1H). Taken together, our
results demonstrate that the one-step generated biallelic mutant pigs developed similar
phenotypes of the human vWD.
A certain level of off-target cleavage by CRISPR/Cas system was suggested by recent
studies in human cell lines
12
, indicating that Cas9-mediated DNA cleavage tolerates small numbers of mismatches
between sgRNA and target DNA especially in protospacer adjacent motif (PAM)-distal
region
12
. We thus sought to test the possible off-target effects in genome-modified pigs derived
from zygote injection. By screening the genome based on sequence homology (> 14 base
pair identity to 3′ end of the sgRNA spacer), around 20 potential off-target sites
were found (Supplementary information, Figure S2). In all 11 mutant animals tested,
none of them contained indels at 2 randomly chosen sites based on T7EI assay (Supplementary
information, Figure S2). We further analyzed the potential off-target effects by Sanger
sequencing of 10 randomly selected potential off-target sites in 3 biallelic mutant
pigs (#9, #10 and #11). None of the sequencing reads had mutations, suggesting no
off-target effects at these sites (Supplementary information, Figure S2). However,
we could not exclude the possible existence of certain off-target effects due to the
high number of potential off-target sites. Thus a systematic off-target evaluation
would be needed in the future before the wide application of such knockout pig models.
Also, modified CRISPR/Cas-methods with less off-target tendency can be used for genome
engineering in pigs and other large animals
13
.
Germline transmission of the mutations to next generation is essential for expanding
the knockout pig population. To examine the germline transmission potential of the
mutations, we dissected an ovary from a monoallelic mutant female pig and a testis
from a biallelic mutant male pig by biopsy and analyzed their genotypes by Sanger
sequencing. Both the ovary and the testis were morphologically normal (Supplementary
information, Figure S3). More than half sequence reads of the ovary and all the sequence
reads of the testis were mutant, indicating that the mutations had been efficiently
transmitted into the germline (Supplementary information, Figure S3). Considering
the highly efficient germline transmission in rodents using the same procedure
14
, germline transmission of the mutations to next generation is highly expected.
In summary, we have shown that zygote injection of the CRISPR/Cas system can efficiently
generate genome-modified pigs in one step, resulting in rapid establishment of large
animal model for important human diseases. The high efficiency of Cas9-mediated genome
editing in pigs demonstrated here, together with the versatile functionality of the
CRISPR/Cas system such as multiplexed genome editing and epigenetic modification
5
, opens up unlimited possibilities of genome engineering in livestock for applications
in agriculture and biomedicine.