- Record: found
- Abstract: found
- Article: not found
The role of high cholesterol in SARS-CoV-2 infectivity.
Read this article at
Abstract
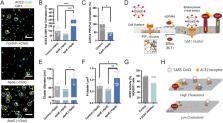
Coronavirus disease 2019 (COVID19) is a respiratory infection caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2). The virus binds to angiotensinogen converting enzyme 2 (ACE2) which mediates viral entry into mammalian cells. COVID19 is notably severe in elderly and those with underlying chronic conditions. The cause of selective severity is not well understood. Here we show cholesterol and the signaling lipid phosphatidyl-inositol 4,5 bisphosphate (PIP 2) regulate viral infectivity through the localization of ACE2’s into nanoscopic (<200 nm) lipid clusters. Uptake of cholesterol into cell membranes (a condition common to chronic disease) causes ACE2 to move from PIP 2 lipids to endocytic ganglioside (GM1) lipids, where the virus is optimally located for viral entry. In mice, age, and high fat diet increase lung tissue cholesterol by up to 40%. And in smokers with chronic disease, cholesterol is elevated two-fold, a magnitude of change that dramatically increases infectivity of virus in cell culture. We conclude increasing the ACE2 location near endocytic lipids increases viral infectivity and may help explain the selective severity of COVID-19 in aged and diseased populations.
Related collections
Most cited references72
- Record: found
- Abstract: found
- Article: not found
SARS-CoV-2 Cell Entry Depends on ACE2 and TMPRSS2 and Is Blocked by a Clinically Proven Protease Inhibitor

- Record: found
- Abstract: found
- Article: found
