- Record: found
- Abstract: found
- Article: found
P2X7 Variants in Oncogenesis
Read this article at
Abstract
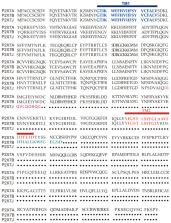
The P2X7 receptor for extracellular ATP is a well-established mediator of tumoral development and progression both in solid cancers and hematological malignancies. The human P2X7 gene is highly polymorphic, and several splice variants of the receptor have been identified in time. P2X7 single-nucleotide polymorphisms (SNPs) have been broadly analyzed by studies relating them to pathologies as different as infectious, inflammatory, nervous, and bone diseases, among which cancer is included. Moreover, in the last years, an increasing number of reports concentrated on P2X7 splice variants’ different roles and their implications in pathological conditions, including oncogenesis. Here, we give an overview of established and recent literature demonstrating a role for human P2X7 gene products in oncological conditions, mainly focusing on current data emerging on P2X7 isoform B and nfP2X7. We explored the role of these and other genetic variants of P2X7 in cancer insurgence, dissemination, and progression, as well as the effect of chemotherapy on isoforms expression. The described literature strongly suggests that P2X7 variants are potential new biomarkers and therapeutical targets in oncological conditions and that their study in carcinogenesis deserves to be further pursued.
Related collections
Most cited references106
- Record: found
- Abstract: found
- Article: not found
Activation of the NLRP3 inflammasome in dendritic cells induces IL-1beta-dependent adaptive immunity against tumors.
- Record: found
- Abstract: found
- Article: not found