- Record: found
- Abstract: found
- Article: found
Epigallocatechin-3-Gallate (EGCG): New Therapeutic Perspectives for Neuroprotection, Aging, and Neuroinflammation for the Modern Age
Read this article at
Abstract
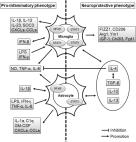
Alzheimer’s and Parkinson’s diseases are the two most common forms of neurodegenerative diseases. The exact etiology of these disorders is not well known; however, environmental, molecular, and genetic influences play a major role in the pathogenesis of these diseases. Using Alzheimer’s disease (AD) as the archetype, the pathological findings include the aggregation of Amyloid Beta (Aβ) peptides, mitochondrial dysfunction, synaptic degradation caused by inflammation, elevated reactive oxygen species (ROS), and cerebrovascular dysregulation. This review highlights the neuroinflammatory and neuroprotective role of epigallocatechin-3-gallate (EGCG): the medicinal component of green tea, a known nutraceutical that has shown promise in modulating AD progression due to its antioxidant, anti-inflammatory, and anti-aging abilities. This report also re-examines the current literature and provides innovative approaches for EGCG to be used as a preventive measure to alleviate AD and other neurodegenerative disorders.
Related collections
Most cited references223
- Record: found
- Abstract: not found
- Article: not found
Alzheimer's disease.
- Record: found
- Abstract: found
- Article: not found
The pro- and anti-inflammatory properties of the cytokine interleukin-6.
- Record: found
- Abstract: found
- Article: found