- Record: found
- Abstract: found
- Article: found
Current Treatment Options and Therapeutic Insights for Gastrointestinal Dysmotility and Functional Gastrointestinal Disorders
Read this article at
Abstract
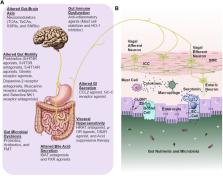
Functional gastrointestinal disorders (FGIDs) have been re-named as disorders of gut-brain interactions. These conditions are not only common in clinical practice, but also in the community. In reference to the Rome IV criteria, the most common FGIDs, include functional dyspepsia (FD) and irritable bowel syndrome (IBS). Additionally, there is substantial overlap of these disorders and other specific gastrointestinal motility disorders, such as gastroparesis. These disorders are heterogeneous and are intertwined with several proposed pathophysiological mechanisms, such as altered gut motility, intestinal barrier dysfunction, gut immune dysfunction, visceral hypersensitivity, altered GI secretion, presence and degree of bile acid malabsorption, microbial dysbiosis, and alterations to the gut-brain axis. The treatment options currently available include lifestyle modifications, dietary and gut microbiota manipulation interventions including fecal microbiota transplantation, prokinetics, antispasmodics, laxatives, and centrally and peripherally acting neuromodulators. However, treatment that targets the pathophysiological mechanisms underlying the symptoms are scanty. Pharmacological agents that are developed based on the cellular and molecular mechanisms underlying pathologies of these disorders might provide the best avenue for future pharmaceutical development. The currently available therapies lack long-term effectiveness and safety for their use to treat motility disorders and FGIDs. Furthermore, the fundamental challenges in treating these disorders should be defined; for instance, 1. Cause and effect cannot be disentangled between symptoms and pathophysiological mechanisms due to current therapies that entail the off-label use of medications to treat symptoms. 2. Despite the knowledge that the microbiota in our gut plays an essential part in maintaining gut health, their exact functions in gut homeostasis are still unclear. What constitutes a healthy microbiome and further, the precise definition of gut microbial dysbiosis is lacking. More comprehensive, large-scale, and longitudinal studies utilizing multi-omics data are needed to dissect the exact contribution of gut microbial alterations in disease pathogenesis. Accordingly, we review the current treatment options, clinical insight on pathophysiology, therapeutic modalities, current challenges, and therapeutic clues for the clinical care and management of functional dyspepsia, gastroparesis, irritable bowel syndrome, functional constipation, and functional diarrhea.
Related collections
Most cited references184

- Record: found
- Abstract: found
- Article: found
The gut-brain axis: interactions between enteric microbiota, central and enteric nervous systems
- Record: found
- Abstract: found
- Article: not found
A Dietary Fiber-Deprived Gut Microbiota Degrades the Colonic Mucus Barrier and Enhances Pathogen Susceptibility.
- Record: found
- Abstract: found
- Article: not found