- Record: found
- Abstract: found
- Article: found
Abiotic production of sugar phosphates and uridine ribonucleoside in aqueous microdroplets
Read this article at
Significance
Phosphorylation is essential for life. Phosphorylated molecules play diverse functions in cells, including metabolic (e.g., sugar phosphates), structural (e.g., phospholipids), and instructional (e.g., RNA and DNA). In nature, the phosphorylation of sugars via condensation is thermodynamically and kinetically unfavorable in bulk solution. Thus, a key question arising within prebiotic chemistry concerning the origin of life is, “How was phosphorus incorporated into the biological world?” Here, we show that sugar phosphates and a ribonucleoside form spontaneously in microdroplets, without enzymes or an external energy source. Sugar phosphorylation in microdroplets has a lower entropic cost than in bulk solution. Therefore, thermodynamic obstacles of prebiotic condensation reactions can be circumvented in microdroplets.
Abstract
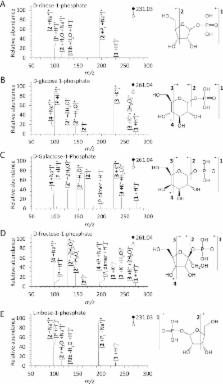
Phosphorylation is an essential chemical reaction for life. This reaction generates fundamental cell components, including building blocks for RNA and DNA, phospholipids for cell walls, and adenosine triphosphate (ATP) for energy storage. However, phosphorylation reactions are thermodynamically unfavorable in solution. Consequently, a long-standing question in prebiotic chemistry is how abiotic phosphorylation occurs in biological compounds. We find that the phosphorylation of various sugars to form sugar-1-phosphates can proceed spontaneously in aqueous microdroplets containing a simple mixture of sugars and phosphoric acid. The yield for d-ribose-1-phosphate reached over 6% at room temperature, giving a Δ G value of −1.1 kcal/mol, much lower than the +5.4 kcal/mol for the reaction in bulk solution. The temperature dependence of the product yield for the phosphorylation in microdroplets revealed a negative enthalpy change (Δ H = −0.9 kcal/mol) and a negligible change of entropy (Δ S = 0.0007 kcal/mol·K). Thus, the spontaneous phosphorylation reaction in microdroplets occurred by overcoming the entropic hurdle of the reaction encountered in bulk solution. Moreover, uridine, a pyrimidine ribonucleoside, is generated in aqueous microdroplets containing d-ribose, phosphoric acid, and uracil, which suggests the possibility that microdroplets could serve as a prebiotic synthetic pathway for ribonucleosides.
Related collections
Most cited references54
- Record: found
- Abstract: not found
- Article: not found
Generalized Gradient Approximation Made Simple.
- Record: found
- Abstract: found
- Article: not found
The protein kinase complement of the human genome.
- Record: found
- Abstract: found
- Article: not found