- Record: found
- Abstract: found
- Article: found
Liquid biopsies to occult brain metastasis
Read this article at
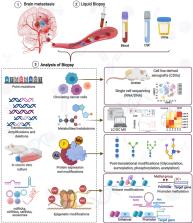
Abstract
Brain metastasis (BrM) is a major problem associated with cancer-related mortality, and currently, no specific biomarkers are available in clinical settings for early detection. Liquid biopsy is widely accepted as a non-invasive method for diagnosing cancer and other diseases. We have reviewed the evidence that shows how the molecular alterations are involved in BrM, majorly from breast cancer (BC), lung cancer (LC), and melanoma, with an inception in how they can be employed for biomarker development. We discussed genetic and epigenetic changes that influence cancer cells to breach the blood-brain barrier (BBB) and help to establish metastatic lesions in the uniquely distinct brain microenvironment. Keeping abreast with the recent breakthroughs in the context of various biomolecules detections and identifications, the circulating tumor cells (CTC), cell-free nucleotides, non-coding RNAs, secretory proteins, and metabolites can be pursued in human body fluids such as blood, serum, cerebrospinal fluid (CSF), and urine to obtain potential candidates for biomarker development. The liquid biopsy-based biomarkers can overlay with current imaging techniques to amplify the signal viable for improving the early detection and treatments of occult BrM.
Related collections
Most cited references201
- Record: found
- Abstract: found
- Article: not found
Inflammation and cancer.
- Record: found
- Abstract: found
- Article: not found