- Record: found
- Abstract: found
- Article: not found
Synthesis of Nucleoside Phosphate and Phosphonate Prodrugs
review-article
Ugo Pradere
† ,
Ethel C. Garnier-Amblard
‡ ,
Steven J. Coats
‡ ,
Franck Amblard
† ,
Raymond F. Schinazi
†
,
21 August 2014
Read this article at
There is no author summary for this article yet. Authors can add summaries to their articles on ScienceOpen to make them more accessible to a non-specialist audience.
Abstract
1
Introduction
For
many decades, the design of new nucleoside analogs as potential
therapeutic agents focused on both sugar and nucleobase modifications.
These nucleoside analogs rely on cellular kinases to undergo stepwise
addition of phosphate groups to form the corresponding active nucleoside
triphosphate to express their therapeutic effect.
1
However, nucleosides triphosphates cannot be considered
as viable drug candidates as they usually have poor chemical stability
along with high polarity that hinders them from transporting across
cell membranes. Within the nucleoside analog phosphate activation
process, the first phosphorylation has often been identified as the
limiting step, which led medicinal chemists to prepare stable “protected”
monophosphate nucleosides capable of delivering nucleoside monophosphates
intracellularly. These nucleoside monophosphate prodrugs are designed
to efficiently cross the biological barriers (as opposed to nucleoside
monophosphates; Figure 1, eq 1) and reach the
targeted cells or tissues. Once inside the cell, the biolabile protecting
groups are then degraded enzymatically and/or chemically, releasing
the free nucleoside analog in the monophosphate form, which can often
efficiently express its therapeutical potency by intracellular conversion
to the corresponding nucleoside triphosphate (Figure 1, eq 2).
Figure 1
Mechanism of action of nucleoside monophosphate prodrugs.
Interestingly, the use of such
phosph(on)ate prodrugs has not only
proved to enhance the activity of parent nucleosides, but also generated
potent compounds otherwise inactive in their nucleoside form because
of a lack of monophosphorylation. Proof of concept for monophosphate
prodrugs has now been clinically validated in the human immunodeficiency
virus (HIV), hepatitis B (HBV), and hepatitis C virus (HCV) fields,
leading to several potent and selective prodrugs such as the phase
II pradefovir,
2
the phase III GS-7340 (TAF),
3
and the FDA-approved tenofovir
disoproxil fumarate (TDF)
4
and sofosbuvir
(GS/PSI-7977) (Figure 2).
5
Figure 2
Examples of clinical nucleoside prodrugs with anti-HIV,
-HBV, or
-HCV activities.
Several strategies allowing
intracellular delivery of nucleotide
analogs were developed over the past 20 years based on the design
of many different types of phosphate and phosphonate nucleoside prodrugs
(Figure 3). Reviews on nucleoside phosph(on)ate
prodrugs generally focus on their enhanced biological activities,
potential therapeutic interest, and their physicochemical properties,
6,7
but almost completely neglect their sometimes challenging synthetic
preparation.
8
Herein, we review the most
important mono-, di-, and triphosphate and phosphonate prodrug approaches
applied to nucleoside analogs (Figure 3) from
a chemical point of view, detailing the strengths and limitations
of each approach. We will focus on the various synthetic pathways
discussing (1) the chemical variation of the biolabile phosph(on)ate
masking groups; (2) the reliability of using P(III) and/or P(V) chemistry
for both phosphate and phosphonate prodrug synthesis; (3) the influence
of the masking group(s) introduction conditions (solvent, temperature,
stoichiometry) on the overall outcome for each method; (4) the various
protection/deprotection strategies used to impart improved yield and
regioselectivity relative to the nature of the nucleobase and the
sugar; and (5) the influence of reaction conditions or protective
groups on the stereoselectivity (R
p/S
p) observed at the phosphorus center as well
as the methods employed to separate both R
p and S
p isomers along with the asymmetric
strategies for the synthesis of predominantly single diastereoisomers
at the phosphorus center.
Figure 3
Prodrug approaches detailed in this Review.
2
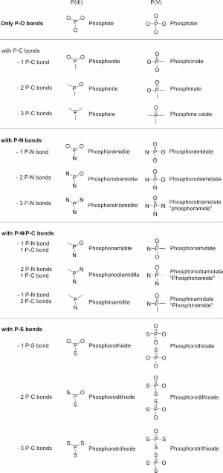
Nomenclature
Phosphorus
is often covalently bonded to common atoms such as C,
H, O, N, S, forming various chemical species or functional groups.
The different categories of phosphorus functional groups are so extensive
that confusion in nomenclature or misused terms is common. As a resource
and useful for an in depth understanding of this Review, Table 1 presents an extensive
summary of the nomenclature
of the principal phosphorus moieties widely used in organic chemistry.
Structures and functional group names are classified by the presence
of O, C, N, and/or S attached to the phosphorus atom and by its valence
(III or V).
Table 1
Functional Group Names of P(III) and
P(V) Moieties
3
Nucleoside
Monophosphate Prodrugs
3.1
Nucleoside Phosphates and
Phosphonates O-PO(OR)2 and C-PO(OR)2
3.1.1
Carbonyloxymethyl (Including
POM, POC)
To date, the only nucleosides phosph(on)ate prodrugs
approved by
the FDA are the acyclic nucleoside phosphonates adefovir dipivoxil
[bis(pivaloyloxymethyl), POM]
9
and tenofovir
disoproxil fumarate [bis(isopropyloxymethyl carbonate, POC].
4
Adefovir dipivoxil was initially developed for
HIV,
10,11
but studies were stopped due to severe kidney
toxicity at the dosage necessary for good antiviral response. In 2002,
further investigation of the compound
12
for the treatment of HBV infection led to FDA approval of adefovir
dipivoxil. The structurally related tenofovir disoproxil fumarate
had a more favorable toxicity profile and was approved in 2001 for
the treatment of HIV infection. Both POM and POC groups have been
shown to increase oral bioavailability
13,14
and overall
systemic exposure to the parent phosphonic acid compound. More recently, LB80380,
a nucleotide bis(POM)-prodrug,
15
completed a phase II clinical trial for the treatment of
lamivudine resistant HBV infection (Figure 4).
16
Figure 4
Examples of carbonyloxymethyl nucleotide
prodrugs approved by the
FDA or in clinical trials.
The degradation of POC-prodrugs involves the enzymatic cleavage
of the carbonate by an esterase leading to an unstable carboxylate
intermediate that undergoes two subsequent chemical degradations to
form carbon dioxide, formaldehyde, and the nucleotide POC-monoester.
Repetition of this sequence with the second POC group or its direct
cleavage by phosphodiesterase frees the nucleoside monophosphate (Figure 5).
Figure 5
Activation of carbonate-type prodrugs (including POC,
R = i-Pr).
In the case of nucleoside prodrugs bearing POM protecting
groups,
the ester is cleaved to form an unstable hydroxymethyl alcoholate
intermediate that undergoes chemical rearrangement to form formaldehyde
and the free monophosphate after the second POM degradation (Figure 6).
Figure 6
Activation of ester-type prodrugs (including POM, R = t-Bu).
The synthetic approaches
for carbonyloxymethyl phosphate nucleoside
prodrugs are summarized in Figure 7: (A) coupling
of a nucleoside monophosphate with a halogeno carbonyloxymethyl derivative
(POM-Cl for example), (B) reaction of a bis(carbonyloxymethyl)-phosphorochloridate
with a nucleoside under basic conditions, (C) Mitsunobu coupling between
a nucleoside and bis(carbonyloxymethyl)-phosphate, and (D) 5′-iodination
of a nucleoside followed by bis(POM)-phosphate salt nucleophilic substitution.
Figure 7
Methods
to access carbonyloxymethyl phosphate nucleosides prodrugs.
The more limited methods to access
carbonyloxymethyl phosphonates
prodrugs are compiled in Figure 8: (A) reaction
of the phosphonic acid nucleoside with an halogeno carbonyloxymethyl
derivative, and (B) direct conversion of dimethylphosphonate nucleoside
using sodium iodide and a halogeno carbonyloxymethyl derivative.
Figure 8
Methods
to access carbonyloxymethyl phosphonates nucleosides prodrugs.
3.1.1.1
Synthesis of Carbonyloxymethyl
Phosphates
Diesters
In 1984, Farquhar and co-workers were first to report
the synthesis of bis(carbonyloxymethyl)phosphate derivatives along
with their stability in different buffers, in the presence of liver
esterase and in plasma.
17,18
They developed two
synthetic routes to synthesize the bis(POM)-monophosphate prodrug
of 5-FdU 2a,b either by Mitsunobu coupling
of (1a,b) with bis(POM)-phosphate 7 or by substitution of a 5-iodo nucleoside 3 with
bis(POM)-phosphate silver salt 8 (Scheme 1). The later method was found to be low
yielding,
and the 3′-acetate could not be removed selectively because
of the lack of POM group stability under deprotection conditions.
Using this approach, numerous nucleosides of biological interest were
transformed in their bis(POM)-monophosphate prodrugs including 5-FdU,
17a,18
2′,3′-dideoxyuridine (ddU),
19
3′-azido-3′-deoxythymidine (AZT),
20
and thymidine.
21
Scheme 1
Synthesis
of 5-FdU Bis(POM)-monophosphate Prodrug
Both phosphates 7 and 8 were
prepared
from common intermediate 6 (Scheme 2), obtained by the reaction between disilver aryl
phosphate 5 and iodomethyl pivalate at room temperature. Hydrogenation
of 6, precipitation as a cyclohexylammonium salt, and
ion exchange on H+-resin provided (7). Subsequent
transformation of bis(POM)-phosphate 7 into its sodium
salt with Na+-resin and final treatment with an aqueous
solution of silver nitrate lead to desired silver salt 8 (Scheme 2).
Scheme 2
Preparation of Reagents 7 and 8
Rose et al.
22
reported the
synthesis
of α/β 2′-deoxy-4′-thioadenosine bis(POM)-monophosphate
prodrug 10 by Mitsunobu coupling between bis(POM)-phosphate 7 and purine nucleosides
9 (Scheme 3).
Scheme 3
Synthesis of 2′-Deoxy-4′-thioadenosine
Bis(POM)-monophosphate
Prodrug
Interestingly, applied
to the synthesis of 8-bromo-2′-deoxyadenosine
bis(POM)-phosphate prodrug, the same method
22
led to an unexpected side reaction of elimination/dehydration, yielding
exocyclic methylene compound 12 (Scheme 4). To circumvent this elimination problem,
nucleoside monophosphate 13 was coupled with commercially available chloromethyl pivalate.
According to the authors, the low yield of 8-bromo-2′-deoxyadenosine
bis(POM)-prodrug 14 (19%) was due to repeated chromatographic
purification.
Scheme 4
Synthesis of 8-Bromo-2′-deoxyadenosine Bis(POM)-phosphate
Prodrug
In 1995, considering
the combersome preparation of bis(POM)-phosphate
nucleoside prodrugs, Imbach et al. developed a new approach allowing
conversion of a nucleoside monophosphate into its corresponding bis(POM)-monophosphate.
23
Thus, AZT monophosphate 15 was
reacted with iodomethyl pivalate and diisopropyl ethylamine in acetonitrile
for 4 days at room temperature to afford AZT bis(POM)-monophosphate
prodrug 16 in 22% yield (Scheme 5).
Scheme 5
Conversion of a Monophosphate into Its Corresponding Bis(POM)-monophosphate
Nucleoside
To increase the reactivity
of the nucleoside monophosphate during
the coupling with POM-I, Kang et al.
24
choose
to preactivate the phosphate moiety as a tributylstannyl salt by using
tributyltin methoxide. As illustrated in Scheme 6, 2′-azido-2′-deoxyuridine monophosphate
17 was first reacted with 2 equiv of tributylstannyl methoxide, then
coupled with iodomethyl pivalate in the presence of tetrabutylammonium
bromide to deliver bis(POM)-prodrug 18 after purification
on reverse phase HPLC. Despite a good overall yield, the use of tin
derivatives represents a serious limitation because of the possible
presence of toxic tin residues incompatible with further biological
evaluations.
Scheme 6
Preactivation of the Phosphate Moiety as a Tributylstannyl
Salt
In 2004, Hwang and Cole developed
a new approach using new bis(POM)-phosphorochloridate 21.
25
This reagent was synthesized
efficiently in five steps from trimethylphosphate by treatment with
sodium iodide and chloromethyl pivalate, monodeprotection, and subsequent
chlorination with oxalyl chloride. The coupling of AZT with bis(POM)-phosphorochloridate
21 in the presence of triethylamine allowed for the formation
of desired AZT bis(POM)-monophosphate prodrug 16 in 47%
yield (Scheme 7).
Scheme 7
Use of Bis(POM)-phosphorochloridate
3.1.1.2
Synthesis
of Carbonyloxymethyl Phosphate
Monoesters
The POM-phosphate monoesters have also been synthesized.
Although these compounds are sometimes evaluated for their biological
activities, they are generally prepared as a reference for metabolic
degradation studies.
Farquhar et al.
18
reported the synthesis of 5-FdU POM-phosphate monoester as a reference
during the degradation study of 5-FdU bis(POM)-prodrug. Starting from
the dibenzyl phosphate silver salt, the POM-protecting group was introduced
by reaction with chloromethyl pivalate. POM-Phosphate 23 was obtained by catalytic
hydrogenation, precipitation of cyclohexylammonium
salts, and neutralization over acidic resin. The coupling between
the dihydrogen POM-phosphate 23 and 5-FdU with DCC in
pyridine afforded POM-5-FdU monophosphate monoester prodrug 2b in 53% yield (Scheme
8).
Scheme 8
Synthesis
of 5-FdU POM-Phosphate Monoester
3.1.1.3
Synthesis of 3′-5′-Cyclic
Carbonyloxymethyl Phosphates
Tsien et al.
26
prepared acetoxymethyl ester prodrugs of N
6,O
2′-dibutyryl adenosine-
and N
2,O
2′-dibutyryl guanosine-3′,5′-cyclic monophosphate, with
the intention of increasing intracellular delivery of second messengers
cAMP and cGMP. The coupling of either diisopropylethylammonium or
silver salts of adenosine-3′,5′-cyclic monophosphate 24 with acetoxymethyl bromide
afforded the acetoxymethyl prodrug
as a mixture of two diastereoisomers 25 and 26 (R
P/S
P)
separated by silica gel chromatography. Interestingly, the diastereomeric
ratio was found to be dramatically different depending on the method
used as the first one afforded a 65:35 mixture in favor of the fast
eluting isomer contrary to the 23:77 mixture obtained with the second
method (Scheme 9). On the other hand, cGMP
prodrug was prepared as a nonseparable mixture of two diastereoisomers
(from derivative X = H) using the DIPEA method.
Scheme 9
Synthesis of N
2,O
2′-Dibutyryl
Adenosine-3′,5′-cyclic Monophosphate
In 2007, Gunic et al.
27
reported the
synthesis of base modified 2′-C-methyl ribonucleosides
cyclic monophosphate prodrugs that exhibited potent anti-HCV activities.
5′-Phosphorylation of nucleosides 27 with POCl3 and P(O)(OMe)3 and subsequent cyclization
using
DCC in pyridine afforded cyclic monophosphate nucleosides 29 in 30% yield (Scheme
10). Finally, coupling
with either chloromethyl pivalate or chloromethyl isopropyl carbonate
in the presence of diisopropylethylamine afforded cyclic POM- and
POC-prodrugs 30 in low to moderate yields (Scheme 10).
Scheme 10
Synthesis of 2′-C-Methyl Ribonucleosides
Cyclic Monophosphates
3.1.1.4
Carbonyloxymethyl Phosphonates
The first synthesis of bis(carbonyloxymethyl)-nucleoside phosphonate
prodrug was reported by Starrett et al.
13,28
who prepared
the bis(POM)-, bis(isobutyryloxymethyl)-, and bis(propionyloxymethyl)-prodrugs
of adefovir (PMEA). At first, the coupling between chloromethyl pivalate
or iodomethyl pivalate and various inorganic (Ag+, Li+, K+, Na+, Cs+) or organic
salts (Et3NH+, (i-Pr)2N+EtH, n-Bu4N+)
of PMEA did not lead to the desired prodrug 31. Finally,
bis(POM)-PMEA was obtained in 40% yield from PMEA by using N,N′-dicyclohexylmorpholine
carboxamidine
(DCMC) as the coupling agent and chloromethyl pivalate. However, the
same procedure was not found suitable for 3-hydroxy-2-phosphonomethoxypropyl
nucleosides such as HPMP-5-azaC, because the reaction lead to an inseparable
mixture of bis(POM)-ester 32 and cyclic POM-monoester
phosphonates 33 (Scheme 11). Optimization
of the reaction conditions (using other salts in place of DCMC, temperature,
and solvents) was not successful.
29
Scheme 11
Difference of Reactivity between PMEA versus HPMP-5-azaC Derivatives
A similar procedure was used
by Choi et al.
15
for the synthesis of 9-[1-phosphonomethoxy
cyclopropyl)methyl]-6-deoxyguanine
dipivoxil LB80380. The nucleoside prodrug was obtained
in two steps by hydrolysis of the diisopropyl phosphonate diester 34 with trimethylsilyl
bromide and coupling of the resulting
phosphonic acid 35 with POM-Cl in the presence of triethylamine
and 1-methyl-2-pyrrolidinone (Scheme 12).
Scheme 12
Synthesis of LB80380
The same procedure was used by Tang et al.
30
to synthesize several PMEA and PMPA bis(alkyloxymethyl)-carbonate
prodrugs. Chloromethyl carbonates 37 were prepared in
60–75% yield from methyl chloroformate, by chlorination with
a large excess of sulfuryl chloride in the presence of catalytic AIBN,
followed by addition of the corresponding alcohol in pyridine. The
coupling of PMEA or PMPA 38 with 4.5 equiv of chloromethyl
carbonates, 37, gave crude 39, which were
converted into their more stable fumarate salts 40 in
50–70% yield (Scheme 13).
Scheme 13
Synthesis
of Several PMEA and PMPA Bis(alkyloxymethyl) Carbonate
Prodrugs
The same procedure
was reported by Mackman et al.
31
to prepare
bis(POC)-5′-phosphonomethoxy prodrugs
of potent nucleosides such as d4T, AZT, ddC, or ddT. Phosphonomethoxy-d4T
and -ddC derivatives were synthesized by electrophilic addition of
dimethyl hydroxymethyl phosphonate to furanoid glycal 41.
32,33
After oxidative deselenylation, deprotection
of the phosphonate moiety and hydrogenation of the double bond, the
resulting phosphonic acid salt 45 was converted to the
bis(POC)-prodrug 46 by coupling with chloromethylisopropyl
carbonate in the presence of triethylamine (Scheme 14).
Scheme 14
Synthesis of Bis(POC)-prodrug 46
This method was later used
for the synthesis of the bis(POC)-5′-phosphonomethoxy
2′-Fd4A prodrug (GS-9148) as shown in Scheme 15.
34,35
Scheme 15
Synthesis of GS9148
To increase the solubility
of highly polar phosphonic acid nucleoside
derivative during coupling reactions and also to reduce the formation
of side-products, lipophilic protecting groups are often temporarily
introduced. Thus, Benzaria et al.
36
reported
the synthesis of bis(POM)-PMEA 51 by protection of PMEA
derivative 48
N
6
-position with a MMTr-group prior to phosphate hydrolysis with TMSBr
and subsequent treatment with triethylammonium bicarbonate (Scheme 16). Finally, the
reaction of compound 50 with iodomethyl pivalate followed by MMTr-deprotection under
acidic
conditions allowed for the bis(POM)-PMEA 51 formation
in 18% yield over two steps.
Scheme 16
N
6-Protection
Prior to Bis(POM)-phosphonate
Nucleoside Formation
MMTr-protection was also employed by Chand and co-workers
to protect
both amine and hydroxyl groups during the synthesis of various C1′-substituted
9-[2-(phosphonomethoxy)ethyl)]adenine
37
and 9-[3-(phosphonomethoxy)propyl]adenine
38−40
bis(POM)- and
bis(POC)-prodrugs derivatives. MMTr-protection of adenosine intermediate 52 and subsequent
selective removal of the pivaloyl group
with NaOH in MeOH afforded compound 54. The phosphonate
moiety was then introduced by coupling with tosylate 55 in the presence of sodium
hydride. The protected dialkyl phosphonate 56 was then hydrolyzed with TMSI in the
presence of triethylamine
to avoid degradation of the MMTr-protecting groups. Finally, the alkylation
of 57 with POM-Cl or POC-Cl proceeded efficiently and
gave the bis(POM)- and bis(POC)-prodrugs 58 in 69–99%
yields, respectively, after deprotection under mild acidic conditions
(Scheme 17).
Scheme 17
N
6- and Hydroxy Group Protection Prior
to Bis(POM)- and Bis(POC)-phosphonates Formation
In 2011, Agrofoglio and co-workers
41
reported the synthesis of 5-substituted uracil
butenyl acyclic bis(POM)-phosphonate
nucleoside 62 by, first, cross-metathesis reaction between
crotylated uracil 60 and dimethyl allylphosphonate 59, followed by direct reaction
with chloromethylpivalate
and sodium iodide (Scheme 18).
Scheme 18
Synthesis
of 5-Substituted Uracil Butenyl Acyclic Bis(POM)-phosphonate
Nucleoside 62
In parallel, the same team developed a more convergent
method for
the synthesis of 5-substituted uracil butenyl acyclic nucleoside bis(POM)-
and bis(POC)-phosphonates 64 and 65 by using
a bis(POM)- or bis(POC)-allylphosphonate as cross-metathesis partner.
42
Bis(POM)- and bis(POC)-allylphosphonates were
generated by reaction of dimethyl allylphosphonate with either POM-Cl
and POC-Cl in the presence of sodium iodide (Scheme 19). Interestingly, the authors
showed that very low conversion
rates were observed when diethylallyl phosphonate was used instead
of dimethyl allylphosphonate. The bis(POM)-prodrugs were finally obtained
after cross metathesis with crotylated uracil 60 using
ruthenium catalyst A at 40 °C. The known instability
of carbonates pushed Agrofoglio’s team to find milder reaction
conditions; thus, the preparation of bis(POC)-prodrugs was achieved
by using IPr indenylidene catalyst B at room temperature
(Scheme 19). A similar procedure was used by
Montagu et al. for the preparation of 5-substituted analogs.
43
Scheme 19
Synthesis of Bis(POM)- and Bis(POC)-allylphosphonates
Nucleoside
Prodrugs
Because of the lack
of reactivity of ruthenium catalysts in the
presence of purines, an alternative strategy was envisaged for the
synthesis of butenyl acyclic purine bis(POM)-phosphonate nucleoside 67–73.
44
Cross-metathesis
between (Z)-2-buten-1,4-diol and bis(POM)-allylphosphonate 63a afforded the desired
(E)-bis(POM)-4-hydroxy-but-2-en-1-yl
phosphonate reagent 66 in 74% yield (Scheme 20). Mitsunobu coupling between 66 and
adenine, 6-chloropurine, or 2-amino-6-chloropurine led to the corresponding
bis(POM)-phosphonate nucleosides 67–69. Further acidic hydrolysis with formic acid
in water gave hypoxanthine 70 and guanine 71 derivatives in 86% and 85%
yields, respectively, while treatment with cyclopropylamine gave 6-cyclopropylamino-
72 and 2-amino-6-cyclopropylamino- 73 derivatives
in 82% and 77% yields, respectively.
Scheme 20
Synthesis of Butenyl
Acyclic Purine Bis(POM)-phosphonate Nucleoside
Prodrugs
More recently, 5′-methylene
phosphonate furanonucleoside
bis(POM)-prodrugs have been prepared through a Horner–Wadsworth–Emmons
reaction between correctly protected 5′-ketal nucleoside intermediates
and a tetra(POM)-bisphosphonate reagent.
45
Uridine, N
4(Boc)2-cytosine, N
6(Boc)2-adenine, 2-N(Boc)2-6-benzyloxy-purine, and 2-N(Boc)2-6-azido-purine 2′-methyl-2′-F-nucleosides
74 underwent oxidation using IBX. Subsequent treatment with
deprotonated tetra(POM)-bisphosphonate reagent 75 afforded
vinyl phosphonate nucleosides 76. TBDMS deprotection
with aqueous formic acid and hydrogenation over palladium hydroxide
afforded the desired prodrugs 77 (Scheme 21).
Scheme 21
Synthesis of 5′-Methylene Phosphonate Furanonucleoside
Bis(POM)-prodrugs
3.1.1.5
Carbonyloxymethyl Phosphonate Monoester
Starrett et al.
13,28
reported the synthesis of PMEA
POM-phosphonate monoester 80. Reaction of diphenyl PMEA 78 with sodium benzoate led
to the unexpected formation of
benzyl monoester PMEA after spontaneous degradation of the dibenzyl
PMEA intermediate. The POM-prodrug 80 was then obtained
by coupling the PMEA benzyloxy monoester 79 with chloromethyl
pivalate in the presence of triethylamine, and subsequent hydrogenation
of the benzyl group with palladium hydroxide on carbon (Scheme 22).
Scheme 22
Synthesis of PMEA POM-Phosphonate Monoester
Prodrug
Tang et al.
30
also reported the synthesis
of PMEA-carbonyloxymethyl monoester 82 by direct coupling
of the phosphonic acid 81 with 1.2 equiv of benzyl or
allyl chloromethyl carbonate in the presence of triethylamine (Scheme 23).
Scheme 23
Synthesis of PMEA-Carbonyloxymethyl Monoester
Prodrug
A similar procedure
was used by Krecmerova et al.
46
for the
synthesis of the 2,6-diaminopurine HPMPC (HPMPC-DAP)
POM-monoester prodrug 84 by reaction of 83 with POM-Cl in the presence of DCMC (Scheme
24).
Scheme 24
Synthesis of (HPMPC-DAP) POM-Monoester Prodrug 84
3.1.1.6
Cyclic
Carbonyloxymethyl Phosphonate
In 2007, Hóly and co-workers
46
reported the synthesis of several cyclic 1-(S)-[3-hydroxy-2-(phosphonomethoxy)propyl]-5-azacytosine
monoester prodrug including POM-derivatives, as an alternative to
the bis(POM)-prodrug. However, instability was found in the HPMP series
due to the presence of a neighboring hydroxyl group. Following Hostetler’s
method,
47
cyclic HPMP-5-azaC was obtained
in quantitative yield by reacting HPMP-5-azaC with dicyclohexylcarbodiimide
and DCMC in dimethylformamide at elevated temperature. cHPMP-5-azaC 85 was converted
to its corresponding tributylammonium salt 86 by treatment with a methanolic solution
of tetrabutylammonium
hydroxide (TBAOH) and finally alkylated with POMCl in dioxane at 100
°C in 52% yield over two steps. Introduction of the POM group
induces the formation of a new chiral center at the phosphorus atom
with a ratio of 5:2 to 3:2 in favor of the trans-isomer 87b (Scheme 25). Only the
cis-isomer 87a was isolated pure on small scale by HPLC
purification. The chair conformation was elucidated by inspection
of spin–spin coupling constants from 1H NMR spectrum.
29
In 2010, Krecmerova et al. took advantage of
this later procedure to prepare the 2,6-diaminopurine cyclic POM-monoester
prodrugs (cHPMP-DAP). The ratio was found to be 6:1 in favor of the
less polar trans-isomer. As before, the diastereoisomers
were distinguished by characteristic values of 31P chemical
shifts, as well as H–H, H–P, and C–P coupling
constants.
Scheme 25
Synthesis of cHPMP-5-azaC POM-Monoester Prodrug
3.1.1.7
Mixed
Prodrugs and Miscellaneous
In 1994, Starrett et al.
13
reported the
synthesis of PMEA mixed glyoxamide POM-diester as part of their pioneering
work on PMEA prodrugs (Scheme 26). Activation
of PMEA with thionyl chloride to form the bis(chloro)-intermediate 88 and subsequent
reaction with N,N-diethylacetamide generated bis(glyoxamide)-PMEA 89. Selective hydrolysis
with sodium hydroxide followed by
alkylation with chloromethyl pivalate in the presence of triethylamine
gave the desired PMEA mixed glyoxamide POM-diester 91.
Scheme 26
Synthesis of PMEA Mixed Glyoxamide POM-Diester Prodrug
In 2007, Fu et al.
48
reported the synthesis
of adefovir bis(l-amino acid)-oxymethyl prodrugs. The desired
adefovir prodrugs 92 were obtained by coupling of N-Boc protected l-amino acid chloromethyl
esters
to PMEA in the presence of DCMC followed by deprotection under acidic
conditions (Scheme 27). Interestingly, these
compounds were found to be 2 times more potent against HBV and 10
times less toxic than adefovir dipivoxil.
Scheme 27
Synthesis of Adefovir
Bis(l-amino acid) Oxymethyl Phosphonate
Prodrugs
3.1.2
S-Acyl-2-thioethyl (SATE)
and S-[(2-Hydroxyethyl)sulfidyl]-2-thioethyl (DTE)
In the early 1990s, a French group first reported that mononucleoside
phosphotriesters, incorporating a thioethyl chain where the thiol
is masked as a thioester (SATE groups, Figure 9), were able to liberate the parent
5′-nucleoside monophosphate
inside the cell.
49
It has been demonstrated
that the decomposition of bis(SATE)-phosphotriester derivatives involves
an esterase-dependent activation process leading to an unstable O-2-mercaptoethylphosphotriester.
This intermediate decomposes
spontaneously via intramolecular nucleophilic displacement into the
corresponding phosphodiester with expulsion of ethylene sulfide (Figure 9). Removal
of the remaining SATE group follows a
similar mechanism giving the desired 5′-O-nucleoside
monophosphate.
50
The same team also reported
a related prodrug containing dithioethanol (DTE) masking groups whose
activation to the same mercaptoethyl intermediate is achieved by a
reductase (Figure 9). The assumed toxicity
concern associated with the ethylene sulfide byproduct has largely
limited the advancement of SATE/DTE prodrugs into development, but
it is commonly used in the in vitro studies to deliver phosph(on)ates
intracellularly.
Figure 9
Activation of (SATE)- or (DTE)-nucleoside prodrugs.
Nucleosides phosph(on)ate prodrug
containing dithioethanol (DTE,
Figure 10) can be prepared by (A) coupling
of bis(DTE)-phosphate intermediate to the nucleoside, and (B) coupling
of a nucleoside phosphonate with 2-substituted (disulfanyl)ethanol
derivatives.
Figure 10
Access to bis(DTE)-phosphotriesters and bis(DTE)-phosphonodiesters.
Bis(SATE)-phosphotriesters nucleosides
can be prepared by (A) coupling
of a H-phosphonate nucleoside with an hydroxythioester
reagent, (B) coupling of a monophosphate nucleoside with an hydroxythioester
derivative, and (C) coupling of a N,N-diisopropylphosphoramidite reagent to a nucleoside
followed by oxidation
of the phosphorus atom (Figure 11).
Figure 11
Access to
bis(SATE)-phosphotriesters and bis(SATE)-phosphonodiesters.
3.1.2.1
Bis(DTE)- and Bis(SATE)-Monophosphate
Historically, the interest for disulfide monophosphate prodrugs
began in the early 1990s with the synthesis and the study of dithioethanol
(DTE) phosphotriester of AZT
23,49
and ddU.
51
Dithioethanol is first monoprotected with a
MMTr group, then phosphorylated with POCl3 to give bis(MMTr-DTE)-phosphate
intermediate 93 in moderate yields. Condensation of either
AZT or ddU with compound 93 led to the corresponding
bis(DTE)-monosphosphate prodrugs 94a and 94b (Scheme 28).
Scheme 28
Synthesis of Bis(DTE)-monosphosphate
Prodrugs
Direct reaction of
nucleosides with bis(SATE)-phosphoramidite is
the most commonly used approach to prepare (SATE)-monophosphate prodrugs.
Lannuzel et al.
52
described the synthesis
of AZT (t-Bu-SATE)-pronucleotide 96 by
first preparing AZT-MP 95. The monophosphate derivative
was then activated by TPSCl and coupled with the S-pivaloyl-2-thioethanol to give
the bis(t-Bu-SATE)-monophosphate
prodrug 96 in good yields (Scheme 29).
Scheme 29
Synthesis of Bis(t-Bu-SATE)-Monophosphate
Prodrug 96
Perigaud et al.
51
reported
the synthesis
of bis(SATE)-ddUMP 99 using H-phosphonate
chemistry (yields not provided). Dideoxy uridine (ddU) was first converted
to the corresponding 5′-hydrogen-phosphonate 97 by reaction with phosphoric acid in
the presence of pivaloyl chloride
and pyridine (Scheme 30). Compound 97 was then reacted with 2-acetylthioethanol 98
upon pivaloyl
chloride activation, to give bis(SATE)-ddUMP 99.
Scheme 30
Synthesis of Bis(MeSATE)-ddUMP Using H-Phosphonate
Chemistry
The most common strategy
to prepare (SATE)-phosphate prodrugs involves
the coupling of a phosphoramidite intermediate 100 with
a nucleoside in the presence of 1H-tetrazole followed
by in situ oxidation with tert-butyl hydroperoxide
or m-CPBA. This method has been successfully applied
to the synthesis of various derivatives of AZT (101),
23
adenallene (102),
53
9-(2′-β-C-methyl-β-d-ribofuranosyl) substituted purines (103, 104),
54,55
pyrrolopyrimidine nucleoside
(105),
56
and IsoddA (106) (Scheme 31).
57
Scheme 31
Traditional (SATE)-Prodrugs Strategies
However, this method has several limitations
related to the nature
of the starting materials. For instance, the presence of exocyclic
amines on the base can lead to competitive substitution and low solubility
of the starting material in commonly used organic solvents.
58
Therefore, bases like G or C have been temporarily
protected with groups such as MMTr or DMTr (Scheme 32).
Scheme 32
Protection of Base Competitive Sites
The presence of a 3′-hydroxy group can
also lead to the
formation of undesired 3′- and 5′,3′-phosphotriester
derivatives. Separation of 3′- and 5′-isomers is not
always straightforward and can require several steps of difficult
chromatographic purification as reported for the synthesis of compound 116 (Scheme
33).
59
In other examples, acid labile protective groups such as
Boc and TBDMS have been used to circumvent the above-mentioned problem
(not shown).
60
Scheme 33
Mixtures with Sugar
Competitive Sites
Ribo nucleosides have also been protected by formation
of a 2′,3′-isopropylidene
group (Scheme 34).
22
Scheme 34
2′,3′-Isopropylidene Group To Mask Competitive
Sites
3.1.2.2
Bis(SATE)-
and Bis(DTE)-Phosphonate
2′-C-Methyl
adenosine phosphonate prodrug
was successfully synthesized by Koh et al.
61
as potential anti-HCV inhibitors. The bis(SATE)-prodrug 127 was found to be slightly
more potent than its phosphonate parent 126b but also more toxic. Starting from 2′-C-methyl
adenosine 123, compound 124 was obtained via a silylation, benzoylation, and desilylation
sequence.
Oxidation of 124 produced the corresponding 5′-aldehyde,
which was subsequently engaged in a Wittig reaction with diphenylphosphoranylidene
methylphosphonate to yield the corresponding 5′,6′-vinyl
phosphonate (not shown). Catalytic hydrogenation of the double bond
followed by transesterification gave the saturated phosphonate ester 125. The 3′-hydroxyl
group was protected with a TBDMS
group followed by removal of the benzoyl group with ammonia and hydrogenolysis
of the benzyl ester to give 3′-protected phophonate 126a. Finally, treatment of 126a
with S-(2-hydroxyethyl)-2,2-dimethylpropanethioate followed by desilylation
lead to desired bis(t-Bu-SATE)-phosphono nucleoside 127 in good yield (Scheme 35,
eq 1).
Interestingly, the authors had to go through this long sequence of
selective protection/deprotection of the 3′-hydroxyl because
direct reaction of S-(2-hydroxyethyl)-2,2-dimethylpropanethioate
with phosphonate 126b in the presence of MSNT yielded
a 3′,5′-cyclic phosphodiester 128 instead
of the desired bis(SATE)-derivative 127 (Scheme 35, eq 2).
Scheme 35
2′-C-Methyl
Adenosine Bis(SATE)-phosphonate
Prodrugs
Benzaria et al.
36,49
also prepared and studied bis(SATE)-
and bis(DTE)-prodrugs of the antiviral agent PMEA 131 (Scheme 36). Hydroxythioesters
precursors
were condensed with N-MMtr-protected PMEA derivative 129 in pyridine in the presence
of 1-mesitylene-2-sulfonyl-3-nitro-1,2,4-triazole
(MSNT) to afford the corresponding phosphonodiesters 130 with monoesters as byproducts.
Finally, deprotection under acidic
conditions provided the target PMEA prodrugs 131.
Scheme 36
Synthesis of Bis(SATE)- or Bis(DTE)-PMEA Prodrugs
Li et al.
62
prepared
the 6′-fluoro-6′-methyl-5′-noradenosine
phosphonic acid bis(SATE)-prodrug 133 by reaction of
phosphonic acid 132 with S-(2-hydroxyethyl)-2,2-dimethylpropanethioate
in the presence of MSNT (Scheme 37).
Scheme 37
Preparation
of Bis(SATE)-Prodrug 133
3.1.2.3
Cyclic Monophosphate Nucleoside Prodrug
Bearing SATE Group
Several cyclic monophosphate (cMP) prodrugs
of heterobase-modified 2′-C-methyl ribonucleoside
were synthesized in 2007 by Gunic et al.
63
Coupling of (29) (refer to Scheme 10 for the synthesis) with appropriate hydroxythioester
in the
presence of MSNT in pyridine gives the corresponding cMP prodrugs 104 and 134 (Scheme
38). Interestingly, (SATE)-cMP prodrugs of nucleosides 29 displayed remarkable improvement
in HCV replicon inhibition (7000–11 000-fold)
without significant toxicity. Activities of these (SATE)-cMP prodrugs
have been shown to be similar to regular 5′-bis(SATE)-MP prodrugs
of nucleosides.
55
Scheme 38
Synthesis of (SATE)-cMP
Prodrugs
In 2010, Liu et al.
64
successfully prepared
3′,5′-cyclic (SATE)-phosphonodiester nucleoside 136 by reacting adenine phosphonic
acid 135 with S-(2-hydroxyethyl)-2,2-dimethylpropanethioate in the presence
of MSNT (Scheme 39).
Scheme 39
3′,5′-Cyclic
(SATE)-Phosphonodiester Nucleoside Synthesis
3.1.2.4
Mixed SATE Approach
3.1.2.4.1
Aryl(SATE)-phosphotriester
The main decomposition pathway
of these aryl (SATE)-phosphotriesters
involves loss of the SATE moiety by action of an esterase, followed
by hydrolysis into the corresponding nucleoside monophosphate through
phosphodiesterase enzymatic activity (Figure 12).
Figure 12
Activation of aryl(SATE)-prodrugs.
Aryl(SATE)-phosphotriesters can be prepared by (A) coupling
of
a N-isopropylphosphoramidite reagent to a nucleoside
followed by oxidation of the phosphorus atom, and (B) coupling of
an already functionalized phosphorochloridate reagent to a nucleoside
(Figure 13).
Figure 13
Methods of preparation of aryl(SATE)-nucleoside
prodrugs.
Villard et al.
65
along with Schlienger
et al.
66
prepared a series of AZT phenyl(SATE)-phosphotriesters
derivatives (Scheme 40). Various thioesters 137 were reacted with phenyldichlorophosphate
to give the
corresponding SATE derivatives 138, which were directly
coupled with AZT in the presence of NMI. Removal of the various protecting
groups was carried out using either TFA or aqueous acetic acid to
provide the desired prodrugs 139 in high yields.
Scheme 40
AZT Phenyl(SATE)-phosphotriesters Prodrugs
Perigaud’s team
67
reported
the
synthesis of (SATE)-phosphotriesters bearing modified l-tyrosinyl
residues by phosphoramidite P(III) chemistry. Condensation of tyrosinyl
precursors with (SATE)-phosphorobis(amidite) reagent 100 led to the corresponding
tyrosinyl(SATE)-phosphoramidite intermediates 140. Reaction of (140) with AZT, followed
by
in situ oxidation with t-BuOOH and treatment of intermediates
under acidic conditions (30% HCl in Et2O or 10% TFAA in
DCM), afforded the desired prodrugs 141 in good overall
yields (Scheme 41).
Scheme 41
Synthesis of (SATE)-Phosphotriesters
Bearing Modified l-Tyrosinyl
Residues
3.1.2.4.2
(SATE)-Phosphoramidate Diester
68
These (SATE)-phosphoramidate diesters containing either simple
aliphatic and aromatic amines or amino acid esters have been shown
to deliver 5′-nucleoside monophosphates after simple esterase
activation followed by phosphoramidase-catalyzed cleavage of the amino
portion (Figure 14).
Figure 14
Activation pathway of
(SATE)-phosphoramidate diester prodrugs.
Perigaud’s research group
68
reported
the synthesis of AZT phosphoramidate diester bearing one (t-Bu)SATE group and various
amino residues using the H-phosphonate chemistry. Thus, key H-phosphonate
monoester 142 was first coupled to AZT in the presence
of t-BuCOCl to give the corresponding (t-Bu)SATE-AZT H-phosphonate diester 143. Finally,
oxidative coupling with various amines afforded the desired
AZT (SATE)-phosphoramidate diesters 144,145 (Scheme 42).
Scheme 42
Synthesis of AZT
(SATE)-Phosphoramidate Diesters Prodrugs
Despite the fact that IDX184 development
for HCV treatment
was stopped in phase IIb in August 2012, the (SATE)-phosphoramidate
diester prodrug of 2′-C-methylguanosine remains
at present the only example of the successful application of this
technology to reach human study. IDX184 was prepared
using the H-phosphonate chemistry similar to that
described above.
69
Thus, the key H-phosphonate monoester precursor was prepared in a few
steps from commercially available 2,2-dimethyl-3-hydroxypropanoic
acid methyl ester, by protection of the alcohol followed by saponification,
leading to compound 146 in 92% yield without purification.
Installation of the side chain was performed by peptidic coupling
between compound 146 and 2-mercaptoethanol to generate
alcohol 147. Finally, treatment of compound 147 with phosphorus acid and pivaloyl
chloride, followed by quenching
the reaction with triethylammonium bicarbonate (TEAB), generated H-phosphonate monoester
precursor 148 in 90%
over two steps. 2′-C-Methylguanosine was then
reacted with 148 in the presence of pivaloyl chloride
to furnish intermediate 149, which was further treated
with benzylamine to generate Tr-protected phosphoramidate diester 150 in quantitative
yield. Classical deprotection with trifluoroacetic
acid led to the isolation of IDX184 in 39% yield (Scheme 43).
Scheme 43
Synthesis of IDX184
3.1.2.4.3
(SATE)-Glucosyl Phosphorothiolates
This type of mixed (SATE)-phosphodiesters
is based on the combination
of the iso(SATE)- and the bis(SGTE)-
70
prodrugs,
two structural modifications previously studied by Perigaud’s
group.
The postulated unmasking pathway of the (SATE)-glucosyl
phosphorothiolate derivatives may involve an esterase activation leading
to the loss of the SATE group and formation of glucosyl phosphorothiolatediesters
(Figure 15). These intermediates should then
undergo a glucosidase-mediated cleavage of the anomeric bond followed
by a rearrangement process similar to the decomposition process proposed
for bis(isoSATE)-pronucleotides.
71
Figure 15
Activation
of (SATE)-glucosyl phosphorothiolate prodrugs.
The synthesis of such (SATE)-glucosyl phosphorothiolate derivatives
involves both P(III) and P(V) intermediates and was developed using
AZT as a model system. The (SATE)-H-phosphonate monoester
precursor 153 was obtained from S-(2-hydroxyethyl)-2,2-dimethylpropanethioate
using salicyl chlorophosphite. Condensation of intermediate 153 with AZT, in the presence
of pivaloyl chloride, led to
the corresponding H-phosphonate diester, which was
in situ oxidized into phosphorothioate 154 using elemental
sulfur (diastereoisomeric mixture 1:1). For the glucosyl phosphorothiolate
portion, a boron trifluoride etherate-induced glycosylation of a pentaacetyl
glucopyranose derivative with 2-bromoethanol gave the corresponding
β-glucopyranoside 151. A Finkelstein halogen-exchange
reaction with sodium iodide was followed by coupling of the resulting
2-iodoethyl-β-d-glucosides 152 with phosphorothioate
diester 154. Phosphorothiolate derivatives 155 were obtained, as a 1:1 diastereomeric
mixture (Scheme 44).
71
Scheme 44
Synthesis of (SATE)-Glucosyl
Phosphorothiolate Derivatives
3.1.2.4.4
(SATE)-Halogeno Phosphodiesters
Egron et al.
72
tried to improve the
anti HIV activity of AZT 5′-fluorophosphate by preparing (t-Bu-SATE)-prodrug 156.
Starting from H-phosphonate diester 143 (refer to Scheme 44 for preparation), fluorination
was achieved using
iodine and triethylamine trishydrofluoride. Pure phosphorofluoridate 156 can be obtained
as a 1:1 mixture of diastereoisomers using
reverse phase column chromatography purification with an isocratic
mixture of acetonitrile in water. It is noteworthy that purification
of compounds 156 on silica gel column chromatography
using MeOH as eluent led to the formation of methylphosphate byproduct 157. However,
this approach was not pursued due to the limited
chemical stability of the (SATE)-phosphorofluoridate diester, which
also provide 158 in buffer media as a side product (Scheme 45).
Scheme 45
Preparation of (t-Bu)SATE
Prodrug 156
3.1.2.4.5
S,S′-Bis(O-acyl-2-oxyethyl)
Phosphorodithiolates: Iso(SATE)-pronucleotides
Schlienger
et al.
70a
studied an isomeric
form of (SATE)-pronucleotides, mononucleoside S,S′-bis(O-acyl-2-oxyethyl) phosphorodithiolates
(iso(SATE)-pronucleotides). The proposed decomposition pathway of
the iso(SATE)-pronucleotides involves: (1) an esterase activation
leading to intermediate A; (2) nucleophilic attack of
the resulting free alcohol on the phosphorus atom, giving rise to
five-membered ring intermediate B; and (3) conversion
of B into 2-mercaptoethylphosphotriester C followed by spontaneous elimination of
episulfide. Removal of the
second iso(SATE) functional group is achieved by a similar mechanism
or by action of phosphodiesterases, allowing the intracellular delivery
of the corresponding nucleoside 5′-monophosphate (Figure 16).
Figure 16
Decomposition pathway of iso(SATE)-nucleoside prodrugs.
Mononucleoside phosphorodithiolates 161 were obtained
in a one-pot procedure involving (pyrrolidino)phosphoramidites 159 and 1H-tetrazole
activation, followed
by oxidation of 160 with t-BuOOH (Scheme 46).
Scheme 46
One-Pot Procedure Involving (Pyrrolidino)phosphoramidites
3.1.3
Cyclosaligenyl
(cycloSal)
Phosphate and Phosphonate Prodrug Approach
cycloSal phosphate and phosphonate prodrugs, originally introduced by
Chris Meier and co-workers, are one the most extensively explored
types of masked nucleotides.
73
This concept
is based on the use of salicylic alcohols to mask the phosphate functional
group of a nucleoside monophosphate (Nu-MP) and has been successfully
applied to the intracellular delivery of a number of antiviral nucleotides
(e.g., AZT, d4T, and acyclovir
74
). Meier’s
research group extensively studied this prodrug and demonstrated that
the intracellular cleavage of cycloSal pronucleotides
is based on an entirely pH-driven chemical hydrolysis mechanism with
no enzymatic activation required. Under basic conditions, the aryl
ester P–O bond is cleaved first, followed by spontaneous cleavage
of the P–O benzyl ester bond (Scheme 47).
Scheme 47
Hydrolysis Pathways of the CycloSal-d4TMP
Triesters
As the cycloSal pronucleotides were designed to
release the active drug via a chemical cascade mechanism, the stability
and hydrolysis pathways of these pronucleotides have been finely tuned
by varying the nature of substituent in the boxed structure (Figure 17). Various diols
were obtained by reduction of
commercially available or prepared salicylic aldehydes, acids, or
esters with NaBH4 or LiAlH4 (Path A). Other
variations were achieved using ortho-formylation
of substituted phenols followed by reduction (Path B) or mild basic
formylation direct hydroxymethylation reactions (Path C). On the other
hand, 7-methylated salicyl alcohols were prepared by alkylation of
their corresponding aldehydes with methyllithium (Path D).
Figure 17
Different
synthetic methods to access cycloSal-diol
precursors.
The coupling of the cycloSal phosphate moiety
to the 5′-hydroxyl group of a nucleoside is achieved using
either P(III) or P(V) chemistry (Figure 18).
However, the strategy using P(III) remains the most common one, due
to the usual lack of reactivity of P(V) species. However, the synthesis
of cycloSal phosphonates is done exclusively through
P(V) chemistry starting directly from nucleoside phosphonates. It
is noteworthy that all of these approaches give no diastereoselectivity
with respect to the configuration at the phosphorus center. Thus,
all compounds are obtained as diastereoisomeric mixtures. Nevertheless,
Meier and co-workers filed a patent
75
reporting
the use of chiral auxiliaries for the synthesis of cycloSal phosphate moieties. Those
species can be separated before coupling
with the nucleoside, leading to diastereomerically pure cycloSal nucleotide prodrugs.
Figure 18
Synthesis of cycloSal prodrugs
via P(III) or P(V)
chemistry.
3.1.3.1
First
Generation
3.1.3.1.1
CycloSal Phosphate
In a general
manner, cycloSal derivative of nucleosides bearing
a pyrimidine base
76
such as AZT
77
or d4T
78
can be easily
obtained using two different methods. In the first approach (Scheme 48), diols 162
are reacted with PCl3 to yield the cyclic intermediate saligenylchlorophosphane 163.
Target molecules 164 are then obtained in
a “one-pot” procedure by coupling nucleosides analogs
with (163), followed by in situ oxidation with t-BuOOH.
Scheme 48
P(III) Chemistry To Access cycloSal
Phosphate Prodrugs
An alternative synthetic approach toward such compounds
involves
the less reactive P(V) chemistry (Scheme 49). The reaction of d4T with phosphorus
oxychloride yields phosphodichloridate 165, which is further reacted with salicyl
alcohol to give
the desired triester 166. However, this last approach
leads to yields remarkably lower (37%) than the one obtained with
the above P(III) approach.
Scheme 49
P(V) Chemistry To Access cycloSal Phosphate Prodrugs
Finally, a third approach
79
has been
used to prepare cycloSal pronucleotides of carbocyclic
nucleoside, the phosphorochloridate chemistry (Scheme 50). Alcohol 167 is first reacted
with phosphorus
oxychloride to give 3-methyl- cyclosaligenylphosphorochloridate 168. Next, reaction
of chlorinated intermediate 168 with nucleoside 169 in pyridine gave the phosphate
triester 170 in 60% yield. However, this method failed
to produce the cycloSal phosphate triester in the
case of the 3′-epi isomer of 169, most likely due to steric hindrance or intramolecular
cyclization.
Scheme 50
Phosphorochloridate Chemistry To Access CycloSal
Phosphate Prodrugs
In contrast to thymidine nucleosides, cytosine derivatives
cause
considerable obstacles when reacted with chlorophosphane 172: for instance, cycloSal
modifications of 3TC or
ddC were achieved in very low yields.
80
In this case, the high reactivity of phosphorus(III) chloride is
counterproductive leading to a mixture of O- and N,O-di-cycloSal derivatives.
To overcome this issue, compound 172 was reacted with
diisopropylamine to give the less reactive phosphoramidite 173 (Scheme 51). This compound
was then selectively
coupled to ddC or 3TC in the presence of pyridinium chloride as an
acid catalyst. Finally, oxidation of phosphite intermediate with t-BuOOH afforded
the corresponding O-cycloSal derivatives 174 and 175 in 75% and 80% yield, respectively.
Scheme 51
Phosphoramidate
Chemistry To Access cycloSal Phosphate
Cytosine Prodrugs
Preparation of cycloSal prodrugs of adenine
or
guanosine nucleotide derivatives by the same method appears more complicated
because of the presence of exocyclic amino groups. However, these
amines can be protected with an acid labile group such as a trityl.
Common base labile protecting groups have to be avoided due to the
potential instability of the target triester derivatives under deprotection
conditions. However, for certain substrates such as ddA and d4A,
81
the preparation of the corresponding cycloSal derivatives was achieved without any
protection
because of the known acid-catalyzed cleavage of the glycosydic bond
of these particular compounds. In the absence of a protective group
on the exocyclic amino group, the regioselective 5′-O-phosphorylation reaction of
ddA and d4A was performed
at −40 °C to enhance the O- versus N-alkylation (8:1 in favor of the 5′-O-modification,
Scheme 52). Using these low
temperature conditions, a 1:1.6 instead of 1:1 diastereoisomeric mixture
was obtained, the later ratio being usually observed for other nucleosides
such as cycloSal-d4TMP.
Scheme 52
Chlorophosphane
Chemistry To Access cycloSal Phosphate
Adenosine Prodrug Derivatives
Spáčilova et al. described the synthesis
of 6-heteroaryl-7-deazapurine
ribonucleosides cycloSal-phosphate pronucleotides 179 as potential adenosine kinase
inhibitors.
82
Interestingly, they demonstrated the relative stability
of the cycloSal prodrug 178 toward Pd-catalyzed
transformations: despite the partial decomposition of the cycloSal phosphate under
basic conditions, Stille and Suzuki
cross-coupling reactions can be performed (Scheme 53). Moreover, like for the protection
of exocyclic amines,
protection of the sugar moiety was achieved by choosing an acid labile
group such as an isopropylidene group that can be easily removed using
90% aqueous trifluoroacetic acid.
Scheme 53
Pd-Catalyzed Reactions with cycloSal Prodrugs
Meier et al.
83
also described
the synthesis
of cycloSal-BVdUMP triesters 183 from
either BVDU 180 or 3′-O-levulinylated
BVdU 181 (Scheme 54), using the
phosphoramidite/oxidation method previously shown in Scheme 51. Interestingly, after
removal of the levulinyl
protection under mild condition by treatment with hydrazine hydrate,
both methods gave similar overall yields (31–50%).
Scheme 54
Deprotection
of a Levulinylate Group on cycloSal-BVdUMP
Triesters
Interestingly, Kortylewicz
et al.
84
prepared
several 5-[125I]iodo-uridine cyclosaligenyl monophosphate
prodrugs for cancer imaging and molecular radiotherapy. Nonradioactive
iodo analogs 185 were prepared by reaction of nucleosides 184 and phosphochloridates
and subsequent oxidation with t-BuOOH. Compounds 185 were then reacted with
hexamethylditin to afford the corresponding 5-trimethylstannyl cycloSal-derivatives,
which were finally engaged in an electrophilic
iodostannylation reaction using Na125I as the source of
radioactive iodine to provide the desired radiolabeled prodrugs 187. Separation of
each diastereoisomers was achieved by reverse
phase HPLC, even though they had close elution profiles (Scheme 55).
Scheme 55
Preparation of Several 5-[125I]Iodo-uridine cycloSal MP Prodrugs
As mentioned earlier, the chirality of the phosphorus
atom leads
to the formation of nucleotide prodrugs as mixtures of two diastereoisomers
(R
p and S
p) in an almost 1:1 ratio. Moreover, the chromatographic separation
of these diastereoisomers, when possible, is often a very difficult
task to achieve.
In 2011, Meier and co-workers
85−87
reported the first synthetic
route to prepare isomerically pure cycloSal-pronucleotides.
Their strategies revolved around the use of chiral auxiliaries that
were introduced by reaction with phosphorus oxychloride followed by
esterification of the resulting dichlorophosphoramidate with salicylic
alcohol. At this stage, their strategy required the facile separation
of the diastereoisomers by chromatography or recrystallization. Final
nucleophilic displacement of the chiral auxiliary by the protected
nucleoside generated diastereomerically pure cycloSal-phosphotriesters, provided that
this reaction took place with
clean inversion of configuration at the phosphorus atom (SNP reaction). Thus, reaction
of (S)-4-isopropylthiazolidine-2-thione
with POCl3 leads to the formation of intermediate 188 that can be further reacted
with 2-(hydroxymethyl)phenol
in the presence of DBU to afford a mixture of two diastereoisomers 189a and 189b.
At this stage, the two compounds
can be separated, and the S
P-configuration
of (189b) was confirmed by X-ray crystallography. The
desired isomer R
P-189a is
coupled with nucleoside 3′-OAc-dT using t-BuMgCl to give access to the diastereomerically
pure monophosphate
prodrug 190 (Scheme 56). The authors
assigned the stereochemistry of the final products, by assuming that
the mechanism of this reaction proceeds with inversion of configuration
at the phosphorus atom.
Scheme 56
Synthesis of Diastereomerically Pure Monophosphate
Prodrug 190
Although the reaction conditions worked well for unsubstituted
salicylic alcohol, the same sequence was surprisingly not applicable
to the synthesis of 3-
85
and 5-
87
methyl-cycloSal derivatives
due to racemization of both the chiral phosphoramidate reagents and
the final nucleoside prodrugs. This led the authors to investigate
the other chiral auxiliaries 191a–e (Scheme 57).
Chiral groups 191 were prepared by reaction of amino
acid derivatives with dimethylcyano dithioiminocarbonate 192. Ultimately, only 191a
and 191e were suitable
for the synthesis of 3- and 5-substituted cycloSal
phosphotriesters because the diastereoisomers were the only ones that
could be separated at the phosphoramidate level. Compounds 191a and 191e were coupled
with cycloSal-phosphochloridates
generating intermediates 193 as a 1:1 mixture of diastereoisomers.
Diastereoisomers R
P-193a and S
P-193b were separated
by chromatography, and the stereochemistry of S
P-193b was confirmed by X-ray crystallography. R
P-193a and S
P-193b, which were more stable than their S
P-and R
P-counterparts,
were coupled with AZT or d4T to form the expected phosphotriesters 194. After nucleophilic
displacement by the nucleoside, both
enantiomerically pure (S
P)- and (R
P)-phosphotriesters 194 could be
isolated. Optimization of the reaction conditions for the third step
was also investigated. Racemization at the phosphorus atom was suppressed
when using [Cu(BEN)](OTf)2 complex in dichloromethane.
Scheme 57
Chiral Auxiliaries for 3- and 5-Substituted CycloSal-Derivatives
It is noteworthy that the authors considered also using
chiral
thiophosphoramidates,
86
but this strategy
failed in the last step as the P=S was not electrophilic enough
to allow for nucleophilic displacement of the chiral auxiliary by
the nucleoside (not shown).
Expending their nucleoside prodrug
research program, Meier and
co-workers reported the development of bis(cycloSal)-pronucleotides
(Figure 19)
88
designed
to deliver two molecules of active drug for each biomolecule administrated.
Figure 19
Meier’s
bis(cycloSal)-pronucleotides.
Conversion of tetrols 197 into corresponding
phosphitylating
agents 200 was realized by treatment with PCl3 under basic condition. Careful control
of the temperature conditions
appeared to be critical to selectively obtain compound 199. Thus, reduction of the
reaction temperature from −20 to
−40 °C helped decrease the quantity of byproducts resulting
from the formation of seven-membered ring 201 (Scheme 58).
Scheme 58
Synthesis of Chlorophosphite 200
The first attempt of coupling
between two molecules of d4T and
crude chlorophosphite 200 led to the targeted pronucleotides 203 after tedious chromatography
and in poor yield (8%, Scheme 59). The synthesis of these bis(cycloSal) compounds
via the phosphoramidite chemistry was also investigated
but did not lead to any improvement in yields. As presented before, cycloSal-pronucleotides
were always obtained as a mixture
of two diastereoisomers (R
P/S
P configuration). In the case of bis(cycloSal)-d4TMPs, two stereogenic centers are
formed in the course of
their preparation. Hence, they should be obtained as a mixture of
three isomers (R
P/R
P, R
P/S
P, and S
P/S
P configuration) in a ratio approaching 1:2:1 depending on the influence
of the nucleoside chirality. In the case of compounds 203, all three diastereoisomers
were isolated close to the expected
1:2:1 ratio. However, according to 1H and 31P NMR spectroscopy, compound 203 was obtained
as a mixture
of three isomers in a ratio of 1:2:2. According to the authors, that
stereodifferentiation may be due to steric interactions between the
two cycloSal-d4TMP units in 3,3′-bis(cycloSal)-d4TMP 203.
Scheme 59
Synthesis of Bis-cycloSal Pronucleotides
Another type of bis(cycloSal)-pronucleotides
was
also developed by Ahmadibeni et al.
89
Thus,
3′-fluoro-3′-deoxythymidine (FLT) and 3′-azido-3′-deoxythymidine
(AZT) bis(cycloSal)-prodrugs 207 were
prepared from tetrol 204 by first formation of bis(chlorophosphite) 205 using PCl3
and then coupling with either AZT
or FLT at low temperature. The subsequent oxidation using t-butyl hydroperoxide (TBHP)
afforded the desired AZT and
FLT bis(cycloSal) derivative as inseparable mixtures
of diastereoisomers (Scheme 60).
Scheme 60
Bis-cycloSal Pronucleotides
3.1.3.1.2
cycloSal Phosphonates
The cycloSal prodrug approach
has also been applied
to the synthesis of phosphonate nucleosides such as PMEA 48 using P(V) chemistry.
First attempts to prepare cycloSal-PMEAs 209, directly from the diethyl ester of PMEA
48, without protection of the exocyclic amino group, led to
a complex mixture of reaction products.
90
To overcome this problem, the diethyl ester exocyclic amino group
was blocked by a monomethoxytrityl protective group (Scheme 61). Treatment of the
intermediate with trimethylsilyl
bromide then gave the bis(trimethylsilyl) ester that was immediately
converted into the corresponding dichloride 208 using
PCl5. The dichloro intermediate 208 was then
reacted with different salicylic alcohols to give the protected cycloSal-PMEA diesters
in low to moderate yields. Finally,
the MMTr group was cleaved by treatment with TFA, which led to the
target cycloSal-PMEAs 209 in 53–82%
yield.
Scheme 61
MMTr Protection/Deprotection To Access cycloSal-PMEAs
Unexpectedly, cycloSal-PMEA derivatives 209 appeared to be
unstable especially in acidic conditions
(pH = 2) and led to the design of possibly more stable cycloaminobenzyl-PMEA
(i.e., cycloAmb-PMEA) phosphoramidates 211.
90
In these molecules, the cycloSal phenolic oxygen atom is replaced by a nitrogen hypothesizing
that the less electronegative nitrogen would reduce the electrophilicity
of the phosphorus atom and consequently increase the stability of
the prodrug. The first attempt to prepare the cycloAmb-PMEAs, using the reaction sequence
shown in Scheme 61, led to the isolation of the targeted compounds 211 (Scheme 62)
in very poor yield
(3–7%). Another approach was then envisaged were PMEA was converted
into its corresponding dichloride derivative 210 by treatment
with oxalyl chloride. Addition of DMF led to the in situ protection
of the nucleobase with a formamidine group. 2-Aminobenzyl alcohols
were then condensed to intermediate 210 to provide corresponding cycloAmb-PMEA derivatives
211 in 25–42%
yield. Interestingly these cycloAmb-PMEA derivatives 211 appeared dramatically more
stable than their cycloSal-PMEA counterparts 209 while still displaying anti-HIV
activity.
Scheme 62
Synthesis of cycloAmb-PMEAs Phosphoramidates
3.1.3.2
“Lock-In” cycloSal-Triesters
Because of the lipophilic character
of cycloSal phosphate triesters and their chemically
triggered
delivery mechanism, a drug concentration equilibrium is generated
across the cell membrane. To trap cycloSal triester
inside the cells and avoid the formation of this equilibrium, so-called
“lock-in” cycloSal pronucleotides were
developed.
91
These triesters are designed
to be enzymatically converted inside the cell into a more polar compound
(Figure 20).
Figure 20
Mechanism of action for “lock-in” cycloSal pronucleotides.
Elaborated acyloxy systems, such as the acetoxymethyl (AM),
isopropyloxycarbonyloxymethyl
(POC), pivaloyloxymethyl (POM),
92
and amino
acid,
93
were used to release the corresponding
carboxylates. Starting from compound 212, obtained using
the standard chlorophosphite procedure, deprotection with TFA led
to cycloSal-d4TMP acid 213. POC and
POM groups can be introduced by reaction of (213) with
the corresponding chloromethyl alkyl reagent to give compounds 214. On the other hand,
a peptidic coupling between (213) and various amino acids leads to the corresponding
amide-containing cycloSal derivatives 215 (Scheme 63).
Scheme 63
Elaborated Acyloxy Systems
Meier et al. developed another type of “lock-in” cycloSal-pronucleotide that bears
a (carboxy)esterase-cleavable
geminal dicarboxylate
91,94
or an acetoxyvinyl
95
group attached to the aromatic ring of the saligenyl
unit. Those new “lock-in” cycloSal-pronucleotides
are enzymatically transformed into a more polar aldehyde or ketone
inside cells (Figure 21).
Figure 21
“Lock-in” cycloSal-pronucleotides
bearing geminal dicarboxylate or acetoxyvinyl groups.
The synthesis of these compounds starts with the
conversion of
4-formylsalicylic alcohols 218 into cycloSal triesters 220 using a standard P(III)-chemistry
route. Next, triesters 220 are reacted with acetic anhydride
and zirconium(IV) chloride to give the corresponding final prodrugs 221 in 23–45%
yield. Interestingly, for some compounds,
a separation of the two diastereoisomers (R
p or S
p) was achieved. The S
p form of the cycloSal triesters demonstrated
improved antiviral activity as compared to the R
p form (Scheme 64).
Scheme 64
Synthesis of “Lock-In” cycloSal-Pronucleotides
Bearing Geminal Dicarboxylate Groups
3.1.4
Cyclic 1-Aryl-1,3-propanyl
Ester HepDirect
HepDirect prodrugs are aryl substituted cyclic
1,3-propanyl esters
developed in the early 2000s by Metabasis Therapeutics, Inc. as a
liver-directed prodrug combining high plasma and tissue stabilities.
So far, three drugs including MB07811(96) and two nucleosides pradefovir
2
and MB07133(97) have been
advanced to human clinical trials (Figure 22). Pradefovir is a 3-chlorophenyl HepDirect
prodrug of Adefovir in
development for hepatitis B infection therapy, while MB07133, a 4-pyridyl HepDirect
prodrug of cytarabine, has been developed
for hepatocellular carcinoma treatment. MB07811 was considered
as a candidate for the treatment of hyperlipidemia.
Figure 22
HepDirect prodrugs in
clinical trial.
These cyclic 1,3-propanyl
esters were designed to undergo oxidative
cleavage catalyzed by the cytochrome P450 (CYP) enzyme 3A, expressed
predominantly in the liver. The hemiketal intermediate can undergo
ring opening to form a negatively charged phosph(on)ate, which subsequently
delivers the free phosph(on)ate nucleoside after spontaneous β-elimination.
The aryl vinyl ketone released during the process of the reaction
is then rapidly detoxified by glutathione S-transferase,
an enzyme present in high concentration in liver cells.
Interestingly,
it was shown that the cleavage of the prodrug portion
depends on the stereochemistry at the benzylic position. Indeed, only
the phosphates with a cis-relationship between the
aryl group and the nucleoside portion (and not the trans) were found to be activated
by CYP3A. In addition, modifications
at the phenyl moiety revealed the importance of an electron-withdrawing
group for sufficient chemical stability (Figure 23).
2b,98
Figure 23
Mechanism of activation for HepDirect
nucleoside prodrugs.
HepDirect phosphate prodrugs can be prepared by coupling
a nucleoside
with a phosphorylating agent derived from a 1-arylpropane-1,3-diol
using either P(III) (diisopropylphospharamidite reagent) or P(V) (nitrophenylphosphate)
chemistry. On the other hand, synthesis of phosphonates is achieved
by direct coupling of 1-arylpropane-1,3-diol with a phosphonate nucleoside
(Figure 24).
Figure 24
Methods to access HepDirect phosphate
or phosphonate nucleoside
prodrugs.
Because HepDirect prodrugs have
two chiral centers (the benzylic
position and the phosphorus atom), nonselective HepDirect prodrug
formation results in the formation of four diastereoisomers. However,
starting from an enantiomerically pure diol results in the formation
of only two diastereoisomers identified as cis and trans that differ only in the configuration
of the newly
formed phosphorus chiral center (Figure 25).
Figure 25
Chirality
in HepDirect prodrugs.
3.1.4.1
Synthesis of Aryl-Substituted Cyclic 1,3-Propanyl
Esters
Enantiomerically pure (R)- and (S)-1-aryl-propane-1,3-diols 223 were obtained
through chromatographic separation of diastereomeric (−)-menthone
ketals (Scheme 65). Alternatively, they can
be synthesized by asymmetric reduction of the aryl ketoacid 224 with (−)- or (+)-B-chlorodiisopinocampheylborane
(DIP-Cl) followed by reduction of the resulting β-hydroxy acid
with LiAlH4 or BH3.Et2O with ee’s
> 96%.
2d,98
Scheme 65
Preparation of Enantiomerically Pure (R)- and (S)-1-Aryl-propane-1,3-diols Using
(−)-Menthone
In certain cases such as 4-pyridyl derivatives, the separation
of diastereoisomers as menthone ketals is difficult, and thus other
chiral moieties have been employed. Esterification of racemic β-hydroxy
ester 225 with N,N-dimethyl
phenylalanine led to an easy separation of both diastereoisomers 226 in high optical
purities and gave the desired diol
S
-226b after removal of the phenylalanate
portion (Scheme 66).
97
Scheme 66
Preparation of Enantiomerically Pure (R)-
and (S)-1-Aryl-propane-1,3-diols Using N,N-Dimethyl-phenylalanine
Enantiomeric enriched (S)-1-(4-pyridyl)-propane-1,3-diol
was also obtained by lipase-mediated resolution in the presence of
porcine pancreatic lipase (PPL) and vinyl acetate in 35–40%
conversion and >95% ee. Final hydrolysis of the acetate groups
led
to compound
S-
226b.
99
3.1.4.2
HepDirect Phosphate
Prodrugs
The first method developed by Erion et al.
98
used P(III) chemistry and the reaction of a
phosphoramidite and
a free nucleoside followed by the oxidation of the phosphate intermediate.
Phosphoramidite 228 is synthesized by reaction of
diol
S
-
223 and commercially available 1,1-dichloro-N,N-diisopropylphosphinamine 227 (Scheme
67). Compound 228 was stable and was
purified by column chromatography on silica gel. The desired HepDirect
prodrug of Lamivudine 229 was obtained as a mixture of cis- and trans-phosphate cyclic
diesters
after coupling of phosphoramidite 228 with 3TC followed
by oxidation with t-BuOOH.
Scheme 67
Synthesis of the
HepDirect Prodrug of Lamivudine
Reddy
et al. used the same phosphite approach to prepare 4-pyridyl
HepDirect prodrug of ara-A.
99
The phosphorylation
step was found to be almost instantaneous at 0 °C, giving a mixture
of cis and trans isomers after oxidation.
However, it was found that the thermal epimerization of the cis–trans mixture (60
°C, 3
h) enables the selective formation of the thermodynamically more stable trans-phosphoramidite.
Finally, the stereospecific oxidation
of P(III) phosphite 231 into P(V) phosphate derivative
resulted in the exclusive formation of trans-HepDirect
phosphate prodrug 232 (Scheme 68).
The stereochemistry of the trans isomer 232 was established using NOE studies, 31P
NMR,
and comparison with similar prodrugs previously reported in the literature.
Scheme 68
Formation of trans-HepDirect Phosphate Prodrug 232
This coupling reaction was also studied
to develop a high throughput
synthesis of HepDirect prodrugs.
100
DMSO
can be also used as a cosolvent when nucleosides are not totally soluble
in DMF (i.e., G nucleosides). The reaction failed to proceed in low
polarity solvents because of the poor solubility of unprotected nucleosides.
Optimization of the stoichiometry of phosphoramidite relative to coupling
agent shows that the best yield (31% ± 14%) can be obtained when
6 equiv of both reagents were used. These conditions were applied
to 148 different nucleosides and show an excessive production of doubly
phosphorylated products. Moreover, the desired monophosphorylated
derivative was only obtained for 52% of the substrates. The use of
2 equiv generally resulted in a decreased yield (11% ± 9%), but
led to a better rate of success with 80% of cases giving the desired
phosphorylated products. The stoichiometry 1:2:2 (nucleoside:phosphoramidate:coupling
agent) is the one generally used for creating nucleoside libraries.
For purification, the most efficient method was determined to be preparative
reverse-phase HPLC with mass-based fraction collection after filtration
of the crude reaction mixture. The process was chosen for its automation
capabilities and ease of HepDirect prodrug preparation. Normal-phase
silica gel cartridge-based purification can also be used but was less
efficient because several sample preparation steps were needed prior
to chromatography. The HPLC purity of these compounds (obtained with
the stoichiometry 1:2:2) was acceptable (90% ± 7%), and the cis–trans ratio was slightly
in favor of the cis-compound.
To obtain the desired cis-isomer prodrugs in a
completely selective manner, Erion et al.
98
developed a chiral p-nitrophenylphosphate reagent
that can react through a SN2-type reaction with a nucleoside.
The p-nitrophenylphosphate trans-isomer can be prepared by reaction of p-nitrophenyl
phosphorodichloridate and 1,3-propanediols to give 234 as a 40:60 cis:trans mixture
of
diastereoisomers (Scheme 69). Interestingly,
stirring the reaction mixture overnight in the presence of an excess
of p-nitrophenol in Et3N shifts the equilibrium
toward the more thermodynamically favored trans-isomer
with less than 3% of cis compound remaining. In the
same manner, the reaction of diisopropylphosphoramidites 233 with p-nitrophenol can
be driven to the exclusive
formation of the single trans-isomer 235 when stirred at room temperature for 8 h.
Subsequent oxidation with t-BuOOH gives access to the desired p-nitrophenylphosphate
trans-isomer 236b.
99
The stereochemistry of the final compounds was determined by NOE
experiments and comparison of benzylic methane proton chemical shift
with literature examples.
Scheme 69
Preparation of Enantiomerically Pure trans p-Nitrophenylphosphates
Unexpectedly,
the coupling of p-nitrophenylphosphate
reagents 236a,b with a free nucleoside in
the presence of bases such as LiH, KOt-Bu, or KNH2 led to the formation of a cis–trans
mixture and/or extensive hydrolysis of the phosphate ester. Surprisingly,
the use of sodium hydride selectively generated the trans-isomer nucleoside prodrug
(in low yield) through an unknown reaction
mechanism. Finally, it was found that the use of t-BuMgCl on 2′,3′-protected nucleosides
resulted in the
exclusive formation of cis-isomers as illustrated
in Scheme 70 with cytarabine (52%)
99
and 2′-Me-A (35%).
101
Determination of the stereochemistry of the final product
was established by comparison of NMR data with literature examples.
Isopropylidene and TBS protective groups were finally removed after
phosphorylation under acidic condition or by treatment with a source
of fluorine (TEAF, TBAF). It is noteworthy that substrates, bearing
leaving groups such as chloro, 4-chlorophenoxy, and 2,4-dichlorophenoxy
groups in place of the nitrophenoxy group, were also tested, but were
found to epimerize during coupling with the nucleoside.
Scheme 70
Formation
of cis-Isomers
Bookser
et al.
102
prepared 3′-amino-3′-deoxyguanosine
monophosphate HepDirect prodrug 242 using temporary protection
of purine 2-NH2 and sugar 3′-hydroxyl with N,N-dibenzylformamidine and TBS groups,
respectively. These protections served two purposes: first, they render
the extremely polar guanosine more manageable in term of solubility
and purification, and they also prevent side reactions. Thus, protected
compound 240 was reacted with p-nitrophenylphosphate
reagent 236a in the presence of t-BuMgCl
to generate corresponding HepDirect intermediate 241 in
93% yield. Finally, treatment with triethylammonium formate (TEAF)
then TFA allowed for the removal of both the formamidine and the TBS
groups. The 2′-N3 group was subsequently reduced
under classical Staudinger reaction conditions to give desired 3′-amino-3′-deoxyguanosine
monophosphate HepDirect prodrug 242 in quantitative yield
(Scheme 71). Determination of the stereochemistry
of the final product was established by comparison of NMR data with
literature examples.
Scheme 71
Synthesis of 3′-Amino-3′-deoxyguanosine
Monophosphate
HepDirect Prodrug
Boyer
et al.
97
also showed that in the
case of cytosine nucleosides, such as ara-C, N
4-protection was necessary to avoid N
4-phosphorylation. Starting from dimethylformamidine derivative 243, coupling with
p-nitrophenylphosphate
reagent 236b in the presence of t-BuMgCl
followed by deprotection under acidic conditions allowed for the preparation
of 4-pyridyl ara-C HepDirect prodrug 244 in gram quantities
(Scheme 72). Unambiguous structural assignement
was made by single-crystal X-ray structure determination of the final
product and confirmed the relative stereochemistry between the aryl
ring and the nucleoside as cis.
Scheme 72
Synthesis of ara-C-HepDirect
Prodrug 244
3.1.4.3
HepDirect Phosphonate Prodrugs
HepDirect phosphonate
prodrugs can be readily prepared from a phosphonic
acid nucleotide. In fact, adefovir HepDirect prodrug was initially
prepared by peptidic coupling conditions of (S)-1-(3-chlorophenyl)-propane-1,3-diol
as a mixture of racemic cis- and trans-isomers (ratio from 55:45 to 60:40, favoring
the cis isomer) separable by chromatography and fractional crystallization
(Scheme 73). Stereochemistry of the cis versus the trans isomers was determined
by 1H and 31P NMR experiments as well as comparison
with known similar examples from the literature.
Scheme 73
Adefovir HepDirect
Phosphonate Prodrugs via Peptidic Coupling Conditions
To favor the formation of the cis-isomers, alternative
coupling procedures and conditions were evaluated. Boyer and collaborators
2d,98
found that nucleophilic substitution at low temperature of an activated
bis-chlorophosphonate 210 led to the formation of 246 in a 75:25 cis:trans ratio (Scheme
74). Finally, the cis-isomer 247 was obtained after deprotection of the imine
group with acetic acid and purification by chromatography in de >95%.
The stereochemistry was assigned on the basis of 1H and 31P NMR and comparison with
the literature, but was ultimately
confirmed by X-ray crystallography.
Scheme 74
Adefovir HepDirect
Phosphonate Prodrugs via a Bis-chlorophosphonate
3.1.5
3′,5′-Cyclic Phosphate Ester
Prodrugs
3′,5′-Cyclic phosphate ester prodrugs
(Figure 26) are part of an interesting prodrug
concept that led to the discovery of PSI-352938, a compound that demonstrated anti-HCV
efficacy in vitro and in
human phase 1 trials. The activation of these derivatives to the monophosphate
involves, first, an enzymatic P–O-dealkylation by CYP3A4 and
then cleavage of the 3′-phosphorus–oxygen bond by phosphodiesterases.
103
Figure 26
Mechanism of activation for 3′,5′-cyclic
phosphate
nucleoside prodrugs.
PSI-352938(104) and
related
analogs were prepared by reacting 6-substituted purine nucleoside 248a with tetra-isopropyl
phosphorodiamidite in the presence
of 4,5-dicyanoimidazole (DCI) and then oxidation to the corresponding cis- and trans-cyclic
phosphate 250 using either I2 or t-BuOOH (Scheme 75). Interestingly, the authors found
that by heating
the mixture of cis and trans phosphite
isomers 249 at 50 °C for several hours, the thermodynamically
more stable intermediate cis-249 was
favored (>95%). It is noteworthy that cis- and trans-phosphate isomers cis-249 and
trans-249 can be isolated by simple
column chromatography and that the structure and stereochemistry of cis-249 was elucidated
using X-ray crystallographic
analysis.
Scheme 75
Synthesis of PSI-3529386 Using P(III)
Chemistry
Yields not provided.
An
alternative approach using P(V) chemistry was developed to stereoselectively
prepare PSI-352938 on multigram scale (Scheme 76). Thus, after optimization of the
reaction conditions,
the desired cis-cyclic phosphate PSI-352938 was obtained as the major isomer by reacting
nucleoside 248b with isopropyldichlorophosphate in the presence of NMI and Et3N.
105
The target compound PSI-352938 was obtained with a purity above 99.5% after either
column chromatography or recrystallization.
Scheme 76
Stereoselective
Synthesis of PSI-352938
3.2
Alkoxyalkyl Monoester (HDP, ODE)
Alkoxyalkyl monoesters
prodrugs, including the hexadecyloxypropyl
(HDP) and octadecyloxyethyl (ODE), are ether lipid phospho-conjugates
(LPC) developed by Hostetler and co-workers in the mid 1990s.
106
This strategy led to the discovery of CMX-001, a HDP prodrug of cidofovir currently
in phase II
clinical trials for CMV and adenovirus infections, and to CMX-157, a HDP prodrug of
adefovir, currently in clinical development for
treatment of HIV infection. Using a similar approach, fozivudine tidoxil,
a thioether lipid prodrug of AZT, reached phase II clinical trials
for the treatment of HIV infection (Figure 27).
Figure 27
Alkoxyalkyl monoester prodrugs in clinical trial.
The concept of these prodrugs is based on the mimicking
of lysophosphatidylcholine
(LPC), a naturally occurring phospholipid. By replacing the choline
moiety by a drug, the prodrug is supposed to use the LPC natural uptake
pathway in the small intestine to reach targeted tissues and achieve
high oral availability. Once delivered into the desired tissue, specific
intracellular enzymes such as phospholipase C cleave the lipid carrier
to free the nucleoside monophosphate (Figure 28).
Figure 28
Metabolism drives the pathway of HDP/ODE prodrugs.
To build a more robust prodrug and prevent undesired
metabolic
reactions, the initial LCP structure was modified over the years.
Thus, the acyl linkage at the sn-1 position of the glycerol backbone
was changed to an ether linkage to prevent cleavage by lysolecithinase,
and the hydroxyl group at the sn-2 position was either reduced or
substituted to prevent a second acylation by acyl transferases. Overall,
alkoxyalkyl monoesters prodrugs, such as HDP, are the result of a
series of chain length, substitutions, and saturation optimizations
(Figure 29).
Figure 29
Modifications of LCP structures.
The synthesis of nucleosides alkoxyalkyl
phosphate prodrugs, summarized
in Figure 30, involves (A) Mitsunobu coupling
between a nucleoside and an alkoxyalkyl phosphate moiety, (B) phosphorylation
of a nucleoside and subsequent introduction of the alkoxyalkyl alcohol,
(C) coupling between a nucleoside and an alkoxyalkyl phosphoramidite
moiety followed by oxidation of the phosphorus atom, and (D) phosphite
condensation and subsequent oxidation.
Figure 30
Methods to synthesize
nucleosides alkoxyalkyl phosphate prodrugs,
PG = 2-chlorophenyl cyanoethyl, AA = alkoxy alkyl.
On the other hand, the synthesis of alkoxyalkyl
phosphonate prodrugs,
shown in Figure 31, has been achieved by (A)
monosaponification of bis(alkoxyalkyl)-nucleoside phosphonate, (B)
direct coupling of a nucleoside phosphonic acid with an alkoxyalkyl
alcohol under Mitsunobu or DCC coupling conditions (strategy employed
in most cases), (C) substitution of monochloro activated nucleoside
phosphonates, (D) direct alkylation of nucleoside phosphonic acid
with alkoxyalkyl halides, and (E) direct introduction of the phosphonate
moiety bearing the alkoxyalkyl chain in a single step by substitution
with a phosphonoalkoxyalkyl oxymethylmethyl tosylate.
Figure 31
Methods to synthesize
nucleosides alkoxyalkyl phosphonate prodrugs,
AA = alkoxy alkyl.
3.2.1
Alkoxyalkyl
Phosphate Monoester Prodrugs
The first synthesis of nucleoside
alkoxyalkyl phosphate monoester
was reported by Piantadosi in 1991 who prepared ether lipid conjugates
monophosphate prodrugs of AZT and ddI.
107
Alkoxyalkyl monophosphates 251 were prepared
by three possible methods (Scheme 77): (1)
by reacting alkoxyalkyl alcohols with diphenyl chlorophosphate followed
by catalytic hydrogenation; (2) by reacting alkoxyalkyl alcohols with
phosphorus oxychloride followed by hydrolysis of the chlorinated intermediate;
or (3) by Arbuzov rearrangement (reaction of an alkoxyalkyl bromide
derivative and trimethylphosphite) and subsequent removal of the methoxy
groups using trimethylsilyl bromide.
Scheme 77
Syntheses of Alkoxyalkyl
Phosphate Derivatives
The
DCC-mediated coupling of the alkoxyalkyl phosphate derivatives 251a–c with AZT afforded
the corresponding
alkoxyalkyl phosphate prodrugs 252 in 22–28% yields
(Scheme 78).
Scheme 78
AZT Alkoxyalkyl
Phosphate Prodrug, AA = Alkoxy Alkyl
Mavromoustakos
et al.
108
demonstrated
that AZT alkoxyalkyl monophosphate prodrugs can be prepared in a more
efficient manner by simply using temporarily protected phosphate derivative 258 (Scheme
79). Thus, starting from
protected glycerol derivative 255, reaction with hexadecyl
bromide followed by acidic removal of the trityl group provided intermediate 256.
Compound 256 was then reacted with o-chlorophenyl phosphodi-1,2,4-triazolide 254 and
treated with triethylamine and water to afford the desired alkoxyalkyl
triethylammonium phosphate salt 257. Finally, the coupling
of AZT with compound 257 in the presence of MSNT was
followed by deprotection with TBAF, which allowed for the preparation
of AZT prodrug 252 in 68% yield.
Scheme 79
AZT Alkoxyalkyl
Monophosphate Prodrugs
In 1997, Hostetler et al.
109
reported
a new chemical approach for the synthesis of octadecyl glycerol (ODG)
and HDP phosphate prodrugs involving the formation of the nucleoside
monophosphate before introduction of the alkyloxyalkyl chain. Thus,
the ODG-acyclovir phosphate prodrug 261 (Scheme 80) was prepared in three steps from
the N
2-acetyl protected acyclovir 259. The phosphate moiety was first introduced on the
protected nucleoside 260 by a DCC-mediated coupling with cyanoethyl phosphate.
Resulting nucleoside cyanoethyl phosphate 260 was subsequently
coupled with 1-octadecyl-glycerol in the presence of MSNT and NMI
in a low 17% yield. Cleavage of the cyanoethyl and N
2-acetyl protective groups with ammonia afforded the desired
prodrug 261 in 92% yield.
Scheme 80
Synthesis of ODG-Acyclovir
Monophosphate Prodrug, MSNT = 1-Mesitylenesulfonyl-3-nitro-1,2,4-triazole
In a similar manner, ODG-AZT was obtained by direct DCC-mediated
coupling of 1-octadecyl-glycerol with AZT monophosphate in 25% yield
(Scheme 81).
Scheme 81
Synthesis of ODG-AZT
Monophosphate Prodrug
Beadle and co-workers
110
reported
the
synthesis of HDP-acyclovir phosphate prodrug 265 by coupling
of 2-chlorophenyl phosphodi-1,2,4-triazolide 254 with N
2-MMTr-protected acyclovir in the presence of
HDPOH and NMI (Scheme 82). The subsequent removal
of the MMTr group with acetic acid was followed by deprotection of
the phenol group under basic conditions to afford the desired acyclovir
HDP phosphate prodrug 265 in 78% yield.
Scheme 82
Synthesis
of HDP-Acyclovir Phosphate Prodrug
A similar procedure was used by Liang et al. in 2006 for
the preparation
of HDP- and ODE-(−)-β-d-(2R,4R)-dioxolane-thymine (DOT) monophosphate prodrug
in 60% yield.
111
The 2-chlorophenyl deprotection
was conducted with a 0.5 N NaOH solution in THF to afford the desired
prodrug in 93% yield (Scheme 83).
Scheme 83
Preparation
of HDP and ODE Dioxolane Prodrugs
Ludwig et al.
112
prepared an
alkoxyalkyl
phosphate monoester prodrug of 5-fluoro-2′-deoxyuridine 272 using P(III) chemistry
(Scheme 84). The alkoxyalkyl hydrogen phosphonate reagent was prepared by reacting
1-O-octadecyl-2-O-acetyl-glycerol
with salicylchlorophosphite in the presence of pyridine followed by
hydrolysis. Reaction of phosphite reagent 270 with 3′-acetyl-5-fluorodeoxyuridine
in the presence of pivaloyl chloride formed the nucleoside hydrogen
phosphonate intermediate 271. Oxidation of P(III) to
P(V) with iodine in water and removal of the acetate group using ammonia
in methanol afforded the desired alkoxyalkyl phosphate prodrug 272 in 82% yield.
Scheme 84
Synthesis of Alkoxyalkyl Phosphate Monoester
Prodrug of 5-Fluoro-2′-deoxyuridine
P(III) chemistry has also been used by Sigmund et al.
113
for the synthesis of AZT and 3′-deoxyadenosine
phospholipid conjugates. 2-(4-Nitrophenyl)ethoxy-protected (NPE) alkoxyalkyl
phosphoramidites 274 were first obtained by reacting
alkoxyalkyl alcohols 273 and diisopropy1amino[2-(4-nitrophenyl)ethoxy]
chloro phosphate. 1H-Tetrazole-mediated coupling
of NPE-protected 3′-deoxyadenosine with phosphoramidite 275 followed by phosphorus
oxidation with iodine afforded
the desired protected prodrugs 276 in excellent yields.
The subsequent deprotection by treatment with DBU in pyridine afforded
the desired prodrug 277 obtained in 71% yield (Scheme 85).
Scheme 85
Synthesis of 3′-Deoxyadenosine Phospholipid
Conjugates, NPE
= 2-(4-Nitrophenyl)ethoxycarbonyl
3.2.2
Alkoxyalkyl Phosphonate Monoester Prodrugs
106a
In 2002, Hostetler et al. reported
the first synthesis of HDP and ODE prodrugs of cyclic and noncyclic
cidofovir. By increasing the oral bioavailability of the parent molecule
cidofovir,
114
its corresponding prodrugs
exhibit increased in vitro antiviral activity against poxviruses,
115
CMV, and other herpes viruses.
116
To extend the alkoxyalkyl prodrug technology
to other HPMP and PME acyclic nucleoside, several efficient syntheses
were developed.
Alkoxyalkyl cidofovir HDP prodrug was initially
obtained by intramolecular cyclization of HPMPC using DCC and DCMC.
The formed cHPMPC salt was then alkylated with alkoxyalkyl bromide
in DMF at 80 °C, which led to HDP cyclic cidofovir prodrug 278 (HDP-cHPMPC) in 33%
yield. The subsequent saponification
allowed for the ring opening and generation of HDP cidofovir prodrug 279 in 58% yield
(Scheme 86).
115
Scheme 86
Synthesis of Alkoxyalkyl Cidofovir HDP
Prodrug
A similar strategy
was later used in an attempt to synthesize 5-aza-HPMPC
117
alkoxyalkyl prodrug 281. 5-Aza-cHPMPC
cyclic phosphonate 86 was synthesized as shown in Scheme 25. This latter compound
was reacted with hexadecyloxyethyl
bromide affording the alkoxyalkyl prodrug in 53% yield as a 3:2 trans/cis ratio at
the newly formed phosphorus
chiral center. Surprisingly, Mitsunobu coupling conditions between
phosphonic acid and alkoxyalkyl alcohol afforded the corresponding
prodrug 280 in only 6.5% yield with recovery of the starting
material. However, the authors were unable to obtain the desired HPMP-5-aza-C
alkoxyalkyl prodrug 281 due to 5-aza-cytosine instability
29
under basic conditions (Scheme 87).
Scheme 87
Synthesis of 5-Aza-HPMPC
This strategy was also used by Krecmerova et al.
46
for the synthesis of the alkoxyalkyl prodrugs
of 2,6-diaminopurine
HPMP analog (HPMPDAP). Pure trans-cHPMPDAP 283 was first obtained by reacting HPMPDAP
with DCC and DCMC.
Conversion of phosphonic acid 283 into its corresponding
tetrabutylammonium salt and subsequent alkylation with hexadecyloxypropyl
bromide afforded the corresponding HDP prodrug 284 in
46% yield as a mixture of trans and cis isomers (1.6:1), partially separable by chromatography.
The stereochemistry
of the cyclic phosphonate 283 and phosphonate esters cis-284 and trans-284 was assigned
on the basis of 1H and 31P NMR
and comparison with the literature. Final saponification of the mixture
of diasteroisomers 284 afforded ring opening and formation
of the HDP-PMPDAP prodrug 285 in 54% yield (Scheme 88).
Scheme 88
Synthesis of HPMPDAP Alkoxyalkyl Prodrugs
An alternative method for the
synthesis of alkoxyalkyl cidofovir
prodrug analogs was developed by Wan et al. and involves a Mitsunobu-type
coupling between cHPMPC 286 and oleyloxypropyl alcohol
in the presence of triphenylphosphine and diisopropyl azadicarboxylate
(DIAD). Ring opening of 287 under basic conditions and
subsequent neutralization with acetic acid afforded the desired OLE-HPMPC
prodrug 288 in 42% yield (Scheme 89).
118
This method was later used for the
synthesis of glycero prodrug derivatives such as 1-O-octadecyl-2-O-benzyl-sn-glycero-3-cidofovir.
This prodrug was shown to target the lungs more specifically.
119
Scheme 89
Synthesis of OLE-HPMPC Prodrug
Valiaeva et al. reported the
preparation of HDP-PMEG prodrug 292.
120
2-Amino-6-chloropurine
phosphonic acid 290 was synthesized
121
by, first, alkylation of diisopropyl 2-chloroethoxymethylphosphonate
with 2-amino-6-chloropurine in the presence of DBU, followed by phosphonate
deprotection with TMSBr. Interestingly, introduction of the alkoxyalkyl
chain was carried out at this stage of the synthesis on the 2-amino-6-chloropurine
nucleoside phosphonic acid, presumably to avoid competitive alkylation
at the O
6-position of guanosine and/or
increase the solubility of the nucleoside. The coupling reaction of
phosphonic acid 290 with HDPOH in the presence of DCC
afforded the corresponding 2-amino-6-chloropurine acyclic nucleoside
phosphonate prodrug 291 in 47% yield. Subsequent acidic
hydrolysis with a 1 M HCl solution and basic neutralization converted
nucleotide 292 into the desired guanosine derivative
in 75% yield (Scheme 90).
Scheme 90
Preparation of HDP-PMEG
Prodrug
This procedure was
also used for the synthesis of phosphonopropoxymethyl
guanine and 2,6-diaminopurine alkoxyalkyl prodrugs, phosphonate isoster
of acyclovir phosphate.
122
Alkylation of
diethyl-3-chloromethoxypropylphosphonate with 2-amino-6-chloropurine
and subsequent deprotection of the phosphonate moiety with TMSBr afforded
2-amino-6-chloropurine nucleoside phosphonic acid 294. The phosphonic acid was converted
into its ODE prodrug 295 as a dimethylamine pyridinium salt, by DCC coupling in the
presence
of DMAP. Subsequent basic hydrolysis with 1 M aqueous sodium hydroxide
and neutralization with acetic acid afforded the ODE-guanosine nucleoside
phosphonate prodrug 296 (Scheme 91).
Scheme 91
Synthesis of Phosphonopropoxymethyl Guanine Alkoxyalkyl Prodrugs
Choo et al. reported the preparation
of the alkoxyalkyl cis-5-phosphono-pent-2-en-1-yl
nucleoside prodrug 299.
123
Uracil diethyl phosphonate
derivative 297 was first synthesized by Mitsunobu coupling
between N
3-benzoylated uracil and (Z)-diethyl (5-hydroxypent-3-en-1-yl)phosphonate followed
by debenzoylation with ammonia in methanol. Triisopropylphenylsulfonylation
and subsequent aminolysis converted the uracil diethylphosphonate 297 into its corresponding
cytosine derivative 298 in 75% yield. Deprotection of the phosphonate moiety with
TMSBr
afforded phosphonic acid, which was subsequently coupled to HDPOH
in the presence of DCC and DMAP (Scheme 92).
Scheme 92
Preparation of Alkoxyalkyl cis-5-Phosphono-pent-2-en-1-yl
Nucleoside Prodrug 299
Beadle et al.
124
developed
an attractive
alternative and more convergent strategy for the synthesis of related
HPMP adenine prodrug 303 based on the introduction of
a phosphonate moiety already bearing the alkoxyalkyl chain. The key
alkoxyalkyl tosylate 301 was synthesized from the diethyl
derivative 300 by, first, deprotection with TMSBr, chlorination
of the resulting phosphonic acid, selective substitution with alkoxyalkyl
alcohol, and, finally, hydrolysis with a saturated NaHCO3 solution. The HPMP-adenine
analog 303 was readily obtained
by alkylation of (S)-9-(3-trityloxy-2-hydroxypropyl)-N
6-trityl-adenine 302 with alkoxyalkyl
toluenesulfonyloxy methylphosphonate 301 followed by
deprotection under acidic condition (Scheme 93).
Scheme 93
Synthesis of HPMP-Adenine Prodrug 303
Alternative preparations of
the related HDP-tosylate 309, through a bis(HDP) P(III)
derivative, have been reported by Vrbkova
et al. (Scheme 94).
125
HDPOH was first treated by PCl3 in pyridine to afford
bis(HDP)-phosphite 304. Standard hydroxymethylation with
paraformaldehyde and triethylamine was followed by tosylation, and
afforded bis(HDP)-tosylate 308 in 25% yield. A more efficient
synthetic pathway to bis(HDP)-tosylate 308 involves bis-activation
of phosphonic acid 307 with oxalyl chloride and subsequent
substitutions with HDPOH. Following a known procedure,
126
monodeprotection by treatment with excess LiN3 in DMF at high temperature led to HDP
hydroxymethyl tosylate 309 in high yield (Scheme 94).
Scheme 94
Preparations of HDP-Tosylate 309
Bis(phosphonomethoxy)-acyclic nucleoside 311 was prepared
in a 60% yield by reacting 310 with 2 equiv of HDP tosylate 301 in DMF. Monosubstituted
compound 312 was
obtained as a side product in 25% yield (Scheme 95).
Scheme 95
Preparation of Bis(phosphonomethoxy)-acyclic Nucleoside 311
Similarly, the 5-fluorocytosine
HPMP derivative 316 was obtained in 69% yield in three
steps: (1) alkylation of the
free alcohol of 313 with HDP tosylate 301, (2) Bz-deprotection by aminolysis, and
(3) trityl removal in acidic
conditions. The direct deprotection of 315 with 80% acetic
acid afforded the 5-fluorouracil derivative in 54% yield (Scheme 96).
120
Scheme 96
Synthesis
of 5-Fluorocytosine HPMP Derivatives 315 and 316
Interestingly, the 2,6-diaminopurine
HPMP derivative could be obtained
following the same procedure, but without nucleobase protection (Scheme 97).
120
Scheme 97
Synthesis
of 2,6-Diaminopurine HPMP Derivative Requires Only Hydroxyl
Protective Group
Using the same key intermediate 301, HDP-PMPDAP
(2,6-diaminopurine)
alkoxyalkyl prodrugs 321a and its 2-amino-6-cyclopropyl
analog 321b were synthesized by Valiaeva et al.
120
Thus, purines were reacted with 1,3-dioxolan-2-one,
and subsequent alkylation with alkoxyalkyl tosylate 301 afforded the desired alkoxyalkyl
prodrugs 321 (Scheme 98).
Scheme 98
Synthesis of HDP-PMPDAP Alkoxyalkyl Prodrugs
and Its 2-Amino-6-cyclopropyl
Analog
In a similar manner,
HDP-PMEDAP, an open ring analog based on the
2,4,6-triaminopyrimidine, was obtained in 19% yield by reaction of
tetrahydropyranyloxyethylamine with 2,4-diamino-6-chloropyrimidine 322 followed by
acidic hydrolysis. Subsequent alkylation of
compound 323 with HDP tosylate 309 afforded
HDP-PMEDAP 324 in 15% yield (Scheme 99).
120
Scheme 99
Synthesis of HDP-PMEDAP
Prodrug
Valiaeva et al.
127
prepared ODE-(S)-MPMP guanosine
compound 327, which was found
to be active against HCV. Compound 327 was easily synthesized
in two steps from the O-benzylated guanosine derivative 325 by first coupling with
alkoxyalkyl tosylate 301 in the presence of t-BuONa, followed by removal
of the benzyl group under acidic conditions (Scheme 100).
Scheme 100
Preparation of ODE-(S)-MPMP Guanosine
Compound 327
Finally, an alternative approach allowing direct conversion
of
the free PME-C, -G, and -A nucleosides into their corresponding alkoxyalkyl
prodrugs was developed by Vrbkova et al.
125
Thus, reaction of PMEG with oxalyl chloride in DMF allowed for the
one-pot chlorination of the phosphorus atom and formation of a formamidine
functional group at the C-2 position. Intermediate 328 was reacted with HDPOH in pyridine
to form a bis(HDP)-substituted
compound. Subsequent deprotection in 80% acetic acid and removal of
one of the alkoxyalkyl chain by treatment with an excess of LiN3 afforded HDP prodrug
330 in high yield (Scheme 101).
Scheme 101
Synthesis of HDP-PMEG Prodrug
Tichy et al.
128
also used a selective
hydrolysis of bis(HDP)-monophosphate derivatives to prepare HDP-(S)-HPMP and HDP-2-(2-phosphonoethoxy)ethyl
(PEE) prodrugs 333 and 336. Thus, starting from bis(isopropyl)-phosphonoethoxyethyl
chloride, treatment with TMSBr, chlorination of the resulting phosphonic
acid, and coupling with HDPO gave the desired bis(HDP)-chloro derivative 331. 2-Amino-6-chloropurine
was then introduced under basic
condition, and the resulting intermediate was hydrolyzed with AcOH
to give the bis(HDP)-guanine prodrug 332. Finally, treatment
with LiN3 in DMF afforded the desired HDP-PEE prodrug 333 (Scheme 102).
Scheme 102
Preparation of
HDP-PEE by Hydrolysis of Bis(HDP)-MP Derivatives
The related HDP-(S)-HPMP prodrug 336 was prepared in six steps from bis(isopropyl)-phosphonoethoxyethyltosylate
306. Thus, 306 was reacted with TMSBr to give
the corresponding phosphonic acid, which was then chlorinated and
reacted with HDPOH. The resulting bis(HDP)-phosphonate 308 was then reacted with 334
in the presence of NaH to
give the bis(HDP)-(S)-HPMPG 335 after
deprotection under acidic conditions. Finally, selective hydrolysis
was achieved by treatment with NaOH in a mixture of dioxane and water
to provide the desired HDP-HPMPG prodrug 336. Interestingly,
the authors also prepared the cyclic monoester 337 by
treatment of 336 with PyBOP in the presence of i-Pr2EtN (Scheme 103).
Scheme 103
Hydrolysis of Bis(HDP)-MP Derivatives To Prepare HDP-(S)-HPMPG
3.2.3
Alkoxyalkyl
Phosphoramidates
Liang
et al.
111
reported the synthesis of alkoxyalkyl
phosphoramidate DOT prodrug 339. This mixed prodrug was
generated by reaction of diphenyl phosphite with DOT, followed by
addition of alkoxyalkyl alcohol. The amino acid portion of the prodrug
was finally introduced by reaction of phosphite intermediate 338 with alanine (Scheme
104).
Scheme 104
Synthesis of Alkoxyalkyl Phosphoramidate DOT Prodrug 339
3.3
Phosphoramidates
and Phosphonamidates O-PO(OR)(NR2) and C-PO(OR)(NR2)
3.3.1
Aryloxy
Amino Acid Amidate ProTide
Aryloxyphosphoramidate prodrugs,
also called “ProTides”,
contain a phosphorus atom bearing an amino acid alkyl ester and an
aryloxy group. Pioneered in the early 1990s by McGuigan and co-workers,
this prodrug approach was the result of several years of SAR studies
during which several types of masked phosphate moieties were evaluated
including bis(alkyloxy)- and haloalkyloxyphosphates,
129
bis(aryloxyphosphate),
130
cyclic
131
and noncyclic alkyloxyphosphoramidates,
129i,132
and phosphorodiamidates.
133
Because
of their ability to increase or even reveal activity of nucleosides,
but also because they are relatively easy to prepare, this type of
prodrug was used in drug discovery settings by medicinal chemists
for the biological evaluation of new nucleosides/tides candidates
in vitro.
134
The proof-of-principle in
humans demonstrated with sofosbuvir (PSI-7977, originally
discovered by Pharmasset, Inc., and approved for the treatment of
HCV) paved the way for the development of several other aryloxyphosphoramidate
prodrugs that have now advanced to clinical trials for HIV treatment
(GS-7340 to phase III,
135
GS-9131 to phase II,
136
stampidine,
to phase I
137
), cancer (thymectacin, in
phase I/II for the treatment of colon cancer
138
), and HCV treatment (INX-08189
139
to phase II, PSI-353661(140) to phase I) (Figure 32).
Figure 32
ProTides
nucleoside in clinical trials or FDA-approved.
The mode of action of these aryloxyphosphoramidates, leading
to
the intracellular delivery of active nucleoside monophosphates, has
been studied in detail over the years.
141
After crossing the cell membrane, the monophosphate deprotection
is initiated by an esterase or cathepsin A producing carboxylate A.
142
A spontaneous intramolecular
cyclization to a five-member ring occurs, releasing a molecule of
phenol. Cyclic intermediate B undergoes chemical opening
in the presence of water leading to phosphoramidate diester C. Finally, cleavage of
C by intracellular phosphoramidase
or histidine triad nucleotide-binding protein 1 (HINT-1)
143
frees the nucleoside monophosphate (Figure 33).
141a,144
Figure 33
Mode of action of aryloxyphosphoramidates/phosphonamidates.
Aryloxyphosphoramidate nucleoside
prodrugs are generally prepared
by three different methods highlighted in Figure 34: (A) coupling of the nucleoside
with a phosphorochloridate
reagent, (B) coupling of a nucleoside with a diarylphosphite and subsequent
oxidative amination, and (C) coupling of an amino acid to a nucleoside
aryl phosphate.
Figure 34
Methods to access phosphoramidates nucleoside prodrugs.
It is noteworthy that these different
synthetic approaches generally
lead to approximate 1:1 mixtures of diastereoisomers at the phosphorus
center often inseparable by flash chromatography. The discovery that S
p and R
p isomers
had different in vitro biological properties lead to the development
of a diastereoselective approach using enantiomerically pure aryloxy
phosphoramidate reagents (Method A′, Figure 34).
On the other hand, the aryloxyphosphonamidate nucleoside
prodrugs
are obtained from the nucleoside phosphonic acid as shown in Figure 35: (A) bis-chlorination
and subsequent substitutions
with phenols and amino acids, (B) DCC coupling with phenols (1 equiv)
followed by chlorination of the nucleoside aryl phosphate and subsequent
substitution with an amino acid, and (C) selective saponification
leading to a nucleoside monoaryl phosphate and subsequent coupling
with an amino acid.
Figure 35
Methods to access phosphonamidates nucleoside prodrugs
(AA = amino
acid).
3.3.1.1
Aryloxy Amino Acid
Phosphoramidate
3.3.1.1.1
Phosphorochloridate
Coupling
Aryloxyphosphoramidate nucleoside prodrugs are generally
prepared
by coupling of nucleosides with phosphorochloridate by either activation
of the imidazolium intermediate with NMI
145
or by 5′-deprotonation of the nucleoside with t-BuMgCl
146
and subsequent substitution
with the chlorophosphoramidate (Figure 36).
147
Figure 36
Mechanism to generate phosphoramidates nucleoside
prodrugs.
Over the past 20 years,
substitution of the phosphorochloridate
reagent has been explored by modifying (1) the nature of the aryloxy
portion (substituted phenols or napthols), (2) the amino acid, and
(3) the amino acid ester. Key phosphorochloridate reagents are generally
prepared by reaction of phosphorus oxychloride with an aryl alcohol
in the presence of triethylamine followed by addition of the appropriate
amino acid alkyl ester.
148
Phosphorochloridates
are generally obtained as a 1:1 mixture of R
p and S
p diastereoisomers.
They are often used crude after a simple extraction or filtration,
but cleaner reaction and higher yields are observed when purified
by a flash chromatography. From all of the natural amino acids, l-alanine is the most
commonly used, while the nature of the
aryl group and carboxyl ester portion is dependent on the nucleoside
and/or its application. In a general manner, the replacement of the
natural amino acid with, for instance, d-amino acids
149
or dialkyl glycine
150
led to significant loss of activity. The only counter-example is
the dimethyl glycine that showed potency almost similar to that of l-alanine.
151
In the same way, any
attempts to replace the amino acid moiety by simple amines
152
or to incorporate methylene linker between
the nitrogen and the ester group of the amino acids
153
led to almost total loss of in vitro activity.
Since
the first use of the NMI-mediated coupling for the synthesis
AZT aryloxyphosphoramidate prodrug by McGuigan et al. in 1992,
130a
numerous nucleoside prodrugs have been successfully
prepared using this approach including AZT,
154,155
d4T,
141b,150,151,156−159
ddU,
160
BVdU,
161
DOT,
111,162
5-trimethylsilyl-arabinofuranosyl
uracil,
163
spiropentane nucleoside,
164
8-aza-isoddA,
165
9-deaza-guanosine,
166
2′-deoxy-2′-fluoro-2′-C-methyl-7-deazapurine,
167
AraU,
168
carbocyclic 2′-methyl-2′-fluoro
uridine,
169
and 2′-C-methyl-6-hydrazinopurine ribonucleoside analogs (Figure 37).
55
On the other
hand, the method employing t-BuMgCl
as a reagent has been successfully employed to prepare d4U and ddU,
170
ddA and d4A,
171
d- and l-carbocyclic d4A and ddA,
172
l-2′-deoxythreofuranosyl 3′-aryloxyphosphoramidate
prodrugs,
173
3TC,
174
l-carbocyclic 2′,3′-dideoxy-2′,3′-didehydro-7-deazaadenosine,
175
2′,5′-dideoxyadenosine,
176
2′,3′-dideoxy-3′-fluoroadenosine,
177
2′-fluoro-6′-methylene-carbocyclic
adenosine,
178
4′-azidouridine,
179
cytidine
180
and
adenosine,
181
and 2′-methyl-4′-azidouridine
and -cytidine
182
prodrugs (Figure 38).
Figure 37
NMI method.
Figure 38
t-BuMgCl method.
These two approaches are substrate-dependent, and therefore
it
is very difficult to predict a priori which one to use for the best
outcome. In some cases, they both provide similar yields like in the
case of 2′-C-methyl-2-amino-6-substituted-purine
ribonucleoside analogs
183
and 5-FdU (Scheme 105).
184
Scheme 105
Comparable
Efficiency between the NMI and t-BuMgCl
Methodologies
In most cases, both
approaches provide the expected product prodrug,
albeit one in better yields than the other as observed by Kumar et
al.
185
during the synthesis of 6-thio-7-deaza-2′-deoxyguanosine
phosphoramidate 364 (Scheme 106, eq 1). Finally, one approach will sometimes afford
the expected
prodrug, while the other one will be completely ineffective. For instance,
McGuigan et al.
186
were unable to synthesize
abacavir and carbovir prudrugs using the NMI-mediated coupling. However,
when 365a and 365b were treated with 1.1
equiv of t-BuMgCl, before adding, respectively, 2.2
and 3 equiv of phosphorochloridate, abacavir was phosphorylated in
43% yield after 36 h, while carbovir was converted to the corresponding
prodrug 366b in 23% yield after 1 week (Scheme 106, eq 2). On the contrary, Yoo et
al.
187
had to use the NMI-approach to prepare 2′-deoxyzebularine
because the treatment of (367) with t-BuMgCl and subsequent addition of phosphorochloridate
never provided
the expected compound 368 (Scheme 106, eq 3).
Scheme 106
NMI versus t-BuMgCl Methodology
These methodologies present some limitations
mainly in the formation
of byproducts that requires, in some cases, the protection of the
nucleobase and/or the sugar moiety.
3.3.1.1.1.1
Byproducts
Reaction efficiency
depends essentially on the presence of the following.
(1) Other
free hydroxyls groups on the sugar backbone of the nucleoside: For
instance, the phosphorylation of unprotected nucleosides bearing competitive
hydroxyl group(s) can lead to a mixture of 5′-mono and 3′,5′-bisphosphorylated
products (often separable by chromatography). However, the same reaction
can also produce, in certain cases, a 3′-monophosphorylated
regioisomer hardly separable from the 5′-phosphorylated product.
5,184b,188
(2) The nature of the
nucleobase: Uridine and adenine nucleoside
analogs can, in general, be phosphorylated with both methods. That
is, no side products resulting from nucleobase phosphorylation are
typically observed for the uridine derivatives, while minor N
6-phosphorylation of adenine can be observed,
but are easily removed during chromatography.
189,190
Cytosine nucleoside prodrugs can be obtained by NMI-mediated coupling;
191
however, the high nucleophilicity of the amine
can lead to partial N
4-phosphorylation,
and therefore the anionic method (t-BuMgCl) is generally
preferred.
174,180
Similarly, conversion of guanosine,
2,6-diaminopurine, and hypoxanthine nucleoside analogs to their corresponding
phosphoramidate prodrugs can often be problematic. Indeed, competitive O
6-phosphorylation can occur, and their low solubility
often limits the efficiency of the reaction. However, both NMI and t-BuMgCl methods
can be used; nevertheless, the NMI-mediated
phosphorylation can fail when solubility of the substrate is very
low.
186
Thus, reaction of allene
derivative 369 with 2.15
equiv of phosphorochloridate in the presence of 4.15 equiv NMI led
to the formation of allenic phosphoramidate 370 along
with a side product presumably identified as the O
6-phosphorylated compound. Interestingly, this bis-phosphorylated
side product was not isolated, because treatment with silica gel and
methanol led to its disappearance. A similar byproduct was observed
during the synthesis of butenol nucleoside prodrug 372, in which the reaction mixture
was treated with 80% AcOH to hydrolyze
the O
6-phosphoramidate prior to chromatography
(Scheme 107).
Scheme 107
Competitive O
6-Phosphorylation, Separation
of Mixtures
Qiu et al. observed
the same phenomena during the synthesis of E- and Z-methylenecyclopropane acyclic
purine nucleoside aryloxyphosphoramidate prodrugs.
190,192
The phosphorylation reaction was performed by treating nucleoside 373(193) with 5
equiv of phosphorochloridate
and 10 equiv of NMI with added pyridine as a solubility enhancing
cosolvent. As a result, the bis-phosphorylated derivate 374 was formed as the major
product. This compound could then be converted
into the desired prodrug 375 in 80% yield after acidic
hydrolysis (Scheme 108).
Scheme 108
Competitive O
6-Phosphorylation, Hydrolysis
to Desired Prodrug 375
3.3.1.1.1.2
Protection of Competitive
Sites on
the Nucleobase
In addition to the use of polar cosolvents
such as pyridine or DMF that can sometimes be beneficial, temporary
protection of the nucleobase or the sugar moiety can alternatively
be used to increase the solubility of the nucleoside and mask other
competitive hydroxy and amino groups.
Thus, Ambrose et al.
194
prepared cytosine methylenecyclopropane acyclic
nucleoside 379 by first N
4-protection of compound 376 with a benzoyl group, followed
by reaction with phosphorochloridate 340 via the NMI-mediated
coupling. In this case, the benzoyl group prevents N
4-phosphorylation reaction but also allows for the separation
of Z and E nucleoside isomers 377a and 377b. Selective benzoyl group deprotection
was carried out by treatment with hydrazine in a 4:1 mixture of pyridine/AcOH
to give the desired prodrug 379 in 29% yield (Scheme 109).
Scheme 109
Competitive N
4-Phosphorylation, Benzoyl
Protection
Because direct phosphorylation
of acyclovir was low-yielding with
the t-BuMgCl method (11%) and ultimately failing
with the NMI-mediated coupling, McGuigan et al. reported the preparation
of acyclovir aryloxyphosphoramidate prodrug 381 from N
2- dimethylformamidine protected nucleoside 382 (Scheme 110).
195
More soluble than the free nucleoside, the DMF-protected
acyclovir was efficiently phosphorylated with the NMI method in 51%
yield. Deprotection in refluxing propanol afforded the desired acyclovir
prodrug in 90% yield. Additional aryloxyphosphoramidate N
2-dimethylformamidine protected analogs were later prepared
with the t-BuMgCl method in yields ranging from 31%
to 93%.
196
However, the deprotection step
usually led to modest yields (2–25%). These low yields were
partially due to the additional HPLC reverse phase purification step
after the classical flash chromatography.
Scheme 110
N
2-Protection as a Formamidine Group
Formamidine protections can also be used to
temporarly protect
the cytidine N
4-exocyclic amine. Thus,
Nilsson et al.
197
synthesized 4′-azido-2′-deoxy-2′-C-methylcytidine prodrug 386 by first reaction
of (383) with dimethylformamide dimethylacetal, followed
by reaction with a chlorophosphoramidate in the presence of NMI and
final deprotection of the amino group under acidic conditions (Scheme 111).
Scheme 111
Competitive N
4-Phosphorylation, Formamidine
Protection
3.3.1.1.1.3
Protection of Competitive Sites on
the Sugar Moiety
Along with the nucleobase protection, temporary
groups can also be introduced on the sugar moiety to increase the
solubility of the starting material and avoid competitive phosphorylation.
For instance, while the direct phosphorylation of 4′-azido
adenosine
181
afforded the desired prodrug 390 in very low yield (6%), the 2′,3′-diol protection
with a cyclopentylidene moiety allowed the coupling of (388) with chlorophosphoramidates
in yields ranging from 71% to 92%.
Mild acidic deprotection using 80% formic acid in water for 4 h afforded
free prodrugs 390 in 47–55% yield (Scheme 112).
Scheme 112
Competitive OH Groups, Protection with
a Cyclopentylidene Moiety
McGuigan et al.
198
protected
nucleoside 391 with an isopropylidene group using a catalytic
amount
of perchloric acid in acetone. Phosphorylation of 392 with 2 equiv of t-BuMgCl and
various phosphorochloridates
afforded the corresponding protected aryloxyphosphoramidate nucleosides
in 30–88% yield. The following deprotection was carried out
in acidic conditions to afford the desired prodrugs 393 in moderate to good yields
(Scheme 113).
Similar alkylidene protection strategies were also employed for the
preparation of several nucleoside analogs such as 5-substituted uridine,
199
4′-azidocytidine
180
and inosine,
200
2′-C-Me-cytidine
201
and adenosine,
198
or ribavirin,
202
using the t-BuMgCl method for the phosphorylation
reaction (not shown).
Scheme 113
Competitive OH Groups, Protection with
an Isopropylidene Moiety
Alternative protecting groups such as benzyloxycarbonyl
(Cbz) have
also proven to be compatible with the synthesis of phosphoramidate
prodrugs. Thus, Cho et al.
203
prepared
Cbz-protected A, U, G, and C derivatives 397 using a
high-yielding three-step process: first 5′-hydroxy TBDMS-protection,
followed by 2′,3′-hydroxyl groups Cbz-protection (along
with the N
4-position in the case of cytosine
derivative), and final TBDMS-removal using Et3N·HF.
The NMI-mediated coupling of 395 with phosphorochloridate 340 afforded corresponding
derivatives in yields ranging from
94% to 98%. Finally, catalytic hydrogenolysis of (396) delivered the desired A, U,
G, and C prodrugs in almost quantitative
yields (Scheme 114). It is noteworthy that
catalytic hydrogen transfer with cyclohexa-1,4-diene and palladium
on charcoal was preferred for uridine derivatives to avoid partial
reduction of the C(5)–C(6) double bond. Despite a long sequence,
the excellent overall yield represents a real improvement to the direct
phosphorylation of unprotected nucleoside (86% to 10% from cytidine,
respectively). This method was eventually applied to the synthesis
of aryloxyphosphoramidate prodrugs of both 2′-deoxy-2′-α-fluoro-2′-β-C-methyl uridine
and cytidine in 87% and 86% yields.
203
Scheme 114
Competitive OH Groups, Cbz-Protection
More recently, Cho et al.
204
showed
that N
6-carbamoyl adenosines nucleosides 399 can also be efficiently coupled with chlorophosphoramidate
340 using t-BuMgCl (Scheme 115).
Scheme 115
NMI, Used as Coupling Agent for the Cbz-Protection
of the Purine
Nucleoside
A temporary levulinate
protecting group has also been reported
by Shen et al. for the synthesis of the vidarabine aryloxyphosphoramidate
prodrug 404 (Scheme 116).
205
Vidarabine was sequentially silylated at the
5′-position and acylated at the 2′- and 3′-positions
with levulinic anhydride. Selective desilylation using TBAF in acetic
acid afforded the correctly protected nucleoside 403 in
87% yield. Interestingly, acetic acid was critical in this reaction
to prevent the levulinyl group from shifting from the 3′- to
the 5′-position. Desired prodrugs were finally obtained by
NMI-mediated phosphorylation and subsequent deprotection with hydrazine
hydrate in a pyridine/acetic acid buffer (Scheme 116).
Scheme 116
Competitive OH Groups, Lev-Protection
Shen et al.
206
also
used a similar sequence
involving temporary protection of compound 405 with levulinates
to prepare the triciribine prodrug 407 (Scheme 117).
Scheme 117
Competitive OH Groups, Lev-Protection
Interestingly, Di Francesco
et al.
207
found the direct formation of
7-substituted phosphoramidate prodrug 410 from the corresponding
parent nucleoside to be problematic
and decided to use tetrahydropyranyl (THP) groups to both protect
the secondary hydroxyl group and the pyrazole moiety. Thus, key intermediate 409 was
obtained in four steps from 408 by 5′-silylation
followed by protection of the 3′-hydroxyl, selective desilylation
using TBAF, and Suzuki coupling with THP-protected pyrazole boronic
acid. Finally, reaction of 408 with chlorophosphoramidate 340 in the presence of t-BuMgCl
followed
by removal of the two THP groups in AcOH afforded the desired 7-substituted
7-deaza-adenine nucleoside prodrug 409 (Scheme 118).
Scheme 118
Synthesis of 7-Substituted 7-Deaza-adenine
Nucleoside Prodrug
In the presence of competitive 5′- and 3′-hydroxyl
groups, it is worth mentioning that reaction conditions can sometimes
be optimized to minimize the formation of undesired species without
the use of protective groups (Scheme 119).
Lehsten et al.
208
reported reaction conditions
for the large-scale synthesis of NB1011, the phosphoramidate
of (E)-5-(2-bromovinyl)-2′-deoxyuridine (BVDU).
They found that the temperature and the rate of addition of the electrophilic
phosphoramidating species were critical factors to selectively phosphorylate
the 5′- over the 3′-hydroxyl groups. The optimized conditions
used dichloromethane as the solvent for the entire process. A ratio
of 1.4:1 for the amino acid HCl salt to PhOP(=O)Cl2 allowed for the optimum formation
of B. Maintaining
the temperature between −10 and 0 °C, a solution of NMI
in dichloromethane was then added dropwise. The reactive mixture is
transferred slowly into a mixture of BVDU in dichloromethane
at −5 °C. The nucleoside, poorly soluble in DCM, slowly
dissolves in the presence of the excess NMI, and this allows further
control of the reaction providing ∼1 kg of NB1011 of high purity (>99% by HPLC) in
53% yield after silica gel chromatography.
Scheme 119
Optimized Reaction Conditions for the Synthesis of NB1011, without the Use of Protective
Groups
3.3.1.1.2
Phosphite Approach
An alternative
approach to the synthesis of aryloxyphosphoramidate nucleoside prodrugs
involves the reaction of a nucleoside with a diaryl phosphite and
subsequent amination under Atherton–Todd conditions with amino
acids.
This method was developed by Li et al. using d4T and
AZT as models.
209
The key diaryl phosphite
was prepared in a two-step procedure involving reaction of phenol
with phosphorus trichloride and subsequent reaction with phosphorous
acid (Scheme 120).
210
The aryloxyphosphoramidate nucleoside prodrugs are then formed in
a one-pot two-step procedure. First, the addition of the nucleoside
to a mixture of 1.5 equiv of diarylphosphite and catalytic amounts
of triethylamine in THF at low temperature quickly yielded the nucleoside
aryl phosphite.
211
Scheme 120
Preparation of
Key Diaryl Phosphite Species
The Atherton–Todd reaction was subsequently carried
out
by addition of amino acid methyl ester, triethylamine, and hexachloroethane.
AZT and d4T prodrugs 413 and 414 were obtained
in 60–72% yield over two steps (Scheme 121, eq 1).
209
Despite this encouraging result,
this reaction appeared limited and was reported later to lead to a
complex mixture of products due to the excess of diaryl phosphite
used to avoid nucleoside dimerization. Ora et al. used the same approach
to prepare thymidine aryloxyphosphoramidate prodrug 415.
212
Thymidine was reacted with diphenyl
phosphite in pyridine and subsequently with alanine methyl ester (yields
not reported, Scheme 121, eq 2).
Scheme 121
Phosphoramidates
via Diaryl Phosphites
Like for the phosphorochloridate coupling, the phosphite
approach
presents some limitations mainly with the formation of byproducts,
which necessitates the protection of the nucleobase and/or the sugar
moiety.
3.3.1.1.2.1
Byproducts
One drawback of
this phosphite approach is the potential formation of complex products
mixtures including dinucleotides and diamino acids species. To overcome
these problems, a different synthetic pathway to the key nucleoside
aryl phosphite intermediate was reported by Jiang et al. based on
P(III) substitution.
213
Using d4T and AZT
as models, this three-component Arbuzov reaction
214
is initiated by reacting the nucleoside with phosphorodichloridate
(1 equiv), t-BuOH, and triethylamine to yield the
corresponding nucleoside aryl phosphate 416 (Scheme 122). These conditions afforded
the nucleoside aryl
phosphite cleanly and in high yield (86% for the p-methoxy phenol derivative). The
addition of 1 equiv of the amino
acid, NCS, and 4 equiv of triethylamine to the solution containing
the intermediate aryl nucleoside 5′-phosphite afforded almost
quantitatively the desired aryloxyphosphoramidate prodrugs 417 in overall yields that
ranged from 63% to 79% over two steps.
Scheme 122
Three-Component Arbuzov Reaction
3.3.1.1.2.2
Protection of Competitive
Sites on
the Nucleobase and/or the Sugar Moiety
The overall yield
of the sequence can usually be improved by masking competitive nucleophilic
sites, which also increases the substrate solubility in commonly utilized
solvents.
For example, Leisvuori et al.
215
prepared 2′-OMe cytidine aryloxyphosphoramidate
prodrug 422 by first 5′-hydroxyl silylation of 418, tritylation of the N
4-position,
and protection of the 3′-hydroxyl with levulinic acid and DCC
in dioxane. Selective 5′-desilylation carried out with TBAF
in a mixture of THF and acetic acid afforded the appropriately protected
nucleoside 420. This latter compound was reacted with
1.2 equiv of diphenyl phosphite in pyridine followed by 1.5 equiv
of alanine methyl ester in the presence of carbon tetrachloride and
triethylamine to afford the protected nucleoside prodrug 421 in 70% yield. Treatment
of 421 with hydrazine, acetic
acid, and pyridine cleaved the levulinoyl group, while the MMTr group
was removed by 80% aqueous AcOH at 65 °C, affording the desired
prodrug 422 in 50% yield (Scheme 123).
Scheme 123
Protection of Competitive Sites – Use of MMTr for the
Nucleobase
and Levulinate for the Sugar
In the same vein, Leisvuori and co-workers
215
used levulinate groups to prepare ribavirin
phosphoramidate
prodrug 426. 2′,3′-Bis-levulinoylated ribavirin 423 was reacted with 1.5 equiv of
diphenyl phosphite in pyridine
to allow formation of the nucleoside phosphite phenyl ester and subsequently
to alanine methyl ester in the presence of carbon tetrachloride and
triethylamine to form the protected nucleoside prodrug 425 in 67% yield. Deprotection
of the levulinoyl groups afforded the
desired ribavirin aryloxyphosphoramidate prodrug 426 in
60% yield (Scheme 124).
Scheme 124
Protection of Competitive
Sites – Levulinate Sugar Potection
In the case of a ribonucleoside, protection of the 2′,3′-diol
with an isopropylidene group can also be envisaged. Thus, Donghi et
al. used this approach for the synthesis of 2′-C-Me-cytidine aryloxyphosphoramidate
prodrugs bearing β-amino
alcohols (Scheme 125).
216
2′-C-Me-cytidine was first protected
with an isopropylidene group after treatment with 2,2-dimethoxypropane
and p-TsOH in acetone in 80% yield. The protected
nucleoside was reacted with diphenyl phosphite in pyridine followed
by amino alcohol to give 429 in 40% yield over two steps.
The final deprotection with TFA in water afforded the desired prodrugs 430 as a mixture
of phosphorus diastereoisomers that were
later separated by HPLC or supercritical fluid chromatography (SFC).
Scheme 125
Protection of Competitive Sites – Isopropylidene Group
3.3.1.1.3
Miscellaneous Approaches
Another
approach involving P(V) chemistry was developed by Nillroth et al.
for the synthesis of FLT-prodrugs.
217
FLT
was first reacted with 2 equiv of o-chlorophenyl
phosphorodichloridate and excess 1,2,4-triazole in the presence of
triethylamine to form the nucleoside aryloxy triazolide phosphoramidate
intermediate 431. The subsequent addition of glycine
methyl ester hydrochloride and triethylamine afforded the FLT-aryloxyphosphoramidate
prodrug 432 in 80% yield (Scheme 126).
Scheme 126
Other Approach Using P(V) Chemistry
In the same study,
217
the use
of a cyclic
phosphorodiamidate reagent for the synthesis of a FLT o-(methynesulfonamino)phenyl
methoxy glycine prodrug analog was also
reported (Scheme 127). Cyclic phosphorochloridate 433 was reacted with glycine methyl
ester in the presence
of triethylamine to form phosphorodiamidate reagent 434. The crude compound was then
directly reacted with FLT, affording
the corresponding FLT-prodrug 435 in 86% yield.
Scheme 127
Use
of Cyclic Phosphorochloridate
Because the direct coupling of dipropylglycine phosphorochloridate
with d4T afforded the desired phosphoramidate prodrug 437 in only 2% yield, McGuigan
et al. designed an alternative approach
for its synthesis.
150
However, the coupling
of d4T 5′-monophenyl phosphate 436(218) with 2 equiv of dipropyl glycine methyl ester
and 2.5 equiv of MSNT in pyridine afforded the desired prodrug 437 in a low 7% yield
(Scheme 128).
Scheme 128
Synthesis of D4T Phosphoramidate Prodrug 437
3.3.1.1.4
Asymmetric Synthesis
It has
been proven over time that S
p and R
p diastereoisomers can display different biological
profiles, and it is not uncommon to see 10-fold or more difference
in terms of in vitro potency between two phosphorus diastereomers.
5,219
The separation of phosphorus diastereomeric mixtures can be realized,
in some cases, by HPLC, selective crystallization, or flash chromatography
on silica gel. However, chemists have more recently developed diastereoselective
approaches based on a phosphorus SN2-type mechanism with
chiral phosphor(odi)amidate reagents.
Thus, Román et
al.
219,220
designed a phosphorodiamidate reagent bearing
a (S)-4-isopropylthiazolidine-2-thione as chiral
auxiliary (Scheme 129). This chiral auxiliary
allows the separation of the S
p and R
p diastereoisomers and acts as a leaving group
during a SN2 reaction with a nucleoside. (S)-4-Isopropylthiazolidine-2-thione was
reacted with phosphoryl chloride
in the presence of triethylamine to give dichlorophosphate 188. The diastereoselective
introduction of the aryl moiety was then
carried out with either DBU or Et3N in acetone at −91
°C. The phosphorochloridates 438 were obtained in
59–93% yields and diastereomeric excess (de) between 28% and
87% depending on the nature of the aromatic moiety. It is noteworthy
that the use of substoichiometric amounts of phenol was required to
avoid formation of diaryl phosphoramidate byproducts that are hardly
separable from the desired product. The introduction of the amino
acid was carried out by reacting the diastereomerically enriched mixture
of phosphorochloridates with a single equivalent of l-alanine
methyl or benzyl ester hydrochloride and 3 equiv of triethylamine
in dichloromethane. Interestingly, the diastereomeric ratios were
found generally lower (∼15–85%) than those for the starting
phosphorochloridate, pointing out a possible isomerization. However,
these phosphorodiamidate reagents 439 can be separated
via flash chromatography to deliver pure diastereoisomers (de >
95%).
Crystals of the major diasteroisomer were obtained, and its structure
and (R)-configuration were confirmed by X-ray crystallography.
Finally, the coupling of d4T with 1 equiv of diastereomerically pure
phosphorodiamidate 439 and 3 equiv of t-BuMgCl in a mixture of THF and pyridine (1:1)
for 5 days at room
temperature afforded the desired prodrugs 440 as single
diastereoisomers in 11–50% yields (85–95% de). The stereochemistry
of the (S
P)-diasteromer was assigned by
comparison with analytical data from the literature.
Scheme 129
Use
of a Chiral Auxiliary
Ross et al.
221
also prepared
several
chiral phosphoramidate reagent bearing substituted phenols that would
act as leaving groups during the phosphorylation step. p-Nitrophenyl phosphoramidate
reagent 441 was prepared
from the commercially available p-nitrophenyl dichlorophosphate
by reaction with phenol and amino acid hydrochloride (Scheme 130). At this stage,
two successive crystallizations
in diisopropyl ether afforded the pure S
p-reagent in 22% yield (96% de). The stereochemistry of the phosphorus
center was assigned by X-ray crystallographic analysis. On the other
hand, pure R
p-isomer was obtained (de
> 99.9%) by purification of the enriched mixture by supercritical
fluid chromatography using a chiral stationary phase. The synthesis
of PSI-7977 was then conducted with the S
p isomer 441 and t-BuMgCl,
affording the desired prodrug 442 in 40% yield (99.7%
de after two recrystallizations from dichloromethane).
Scheme 130
Crystallization
of Phosphoramidate Reagent
In the same paper, the authors also investigated the influence
of other electronegatively substituted phenol moieties (nitro groups
and halogens). 2,4-Dinitrophenol and pentafluorophenol phosphoramidates
were found to be the most reactive reagents. The 2,4-dinitrophenol
phosphoramidate had low selectivity between 3′- and 5′-hydroxy
groups, leading to a higher proportion of 3′,5′-bis(phosphorylated)-nucleoside,
whereas the pentafluorophenyl reagent was more selective and therefore
was selected in further studies.
Compound 443 was
prepared by reaction of phenyl dichlorophosphate
with l-alanine isopropyl ester hydrochloride followed by
pentafluorophenol addition (Scheme 131). After
filtration of the salts, the crude solid was triturated in a mixture
of 20% ethyl acetate in hexanes solubilizing only the desired S
p isomer (de >98%). The stereochemistry of
(S
P)-443 was determined by
X-ray
crystallography. Coupling conditions of (443) with 2′-Me,2′F-nucleoside
were optimized, and it was found that low temperature of reaction
(−5 °C) and slow addition of reagent lowered the formation
of both 3′-phosphorylated and 3′,5′-bisphosphorylated
side products while maximizing reaction conversion. Finally, the multigram
scale synthesis of PSI-7977 was carried out by treating
2′-C-Me-2′-F-nucleoside with 2.1 equiv
of t-BuMgCl in THF at −5 °C followed
by the addition of 1.2 equiv of pentafluorophenol phosphoramidate 443. After being
stirred for 18 h at 5 °C and two successive
crystallizations, PSI-7977 was obtained in an excellent
68% yield (de > 99.7%).
Scheme 131
Synthesis of PSI-7977 (Sofosbuvir)
3.3.1.1.5
Post Modifications of Phosphoramidate
Nucleoside Prodrugs
Interestingly, aryloxyphosphoramidate
nucleoside prodrugs have proven to be stable enough to undergo further
modifications. Thus, Velázquez et al. prepared AZT, d4T, and
thymidine heterodimers with TSAO-T as potential inhibitors of HIV-1
reverse transcriptase.
222
Thymidine aryloxyphosphoramidate
prodrug formation was performed using 2 equiv of phosphorochloridate 340 and 6 equiv
of NMI (Scheme 132).
The corresponding nucleoside phosphoramidate underwent N
3-alkylation with 1,3-dibromopropane, and 444 was then coupled to TSAO-T using potassium
carbonate. Desired heterodimer 446 was obtained in 81% yield.
Scheme 132
Post Modifications
– Alkylation Reactions
As seen previously for the removal of Cbz groups, hydrogenation
conditions are compatible with the aryloxyphosphoramidate moiety.
Thus, reduction of l-Cd4A prodrug 447 gave the
corresponding l-ddA phosphoramidate 449 in 49%
yield (Scheme 133).
172
Scheme 133
Hydrogenation of l-Cd4A to l-ddA phosphoramidite
Similarly, Hatton et al. reported
the reduction of the 4′-C-3′-O-propylene-linked bicyclic
pyrimidine nucleoside (Scheme 134).
223
While the cytosine analog 450a was hydrogenated with hydrogen over Pd/C, the uracil
derivative 450b was hydrogenated by transfer hydrogenation over Pd/C
to minimize the simultaneous uracil base hydrogenation.
Scheme 134
Post
Modifications – Hydrogenation Reactions
Postmodification of phosphoramidate nucleoside
prodrugs by palladium-catalyzed
reactions has also been reported by Perlikova et al.
224
(Scheme 135). 6-Chloro-7-deazapurine
ribonucleoside was first protected with an isopropylidene group before
reaction with t-BuMgCl and phosphorochloridates.
Phenyl, furyl, thienyl, and dibenzofuryl groups were then introduced
at the 6-position using Suzuki–Miyaura or Stille cross-coupling
reactions. Finally, isopropylidene deprotection with 90% TFA at room
temperature led to the desired aryloxyphosphoramidate prodrugs 454 in yields ranging
from 40% to 87%. It is noteworthy that
partial hydrolysis of aryloxyphosphoramidate ester group was observed
during the deprotection reaction.
Scheme 135
Post Modifications
– Palladium-Catalyzed Cross-Coupling Reactions
3.3.1.2
Aryloxy Amino Acid
Phosphonamidate
Despite its long running success with regular
phosphate nucleosides,
ProTide technology has not been widely exploited with phosphonate
nucleoside until recently.
One method developed by Ballatore
et al. involves the bis-chlorination of the phosphonic acid nucleoside
(PMPA) with thionyl chloride and subsequent substitutions with phenol
and l-alanine methyl ester in the presence of triethylamine.
225
Interestingly, the nucleoside was reacted again
with thionyl chloride between the two substitutions, presumably to
reactivate the potential hydrolyzed product. In these conditions,
PMPA-aryloxyphosphonamidate prodrug 456 was only obtained
in 5% yield (Scheme 136).
Scheme 136
Synthesis of PMPA-Aryloxy
Phosphonamidate Prodrug
A more efficient method was developed by Chapman et al.
for the
kilogram scale synthesis of GS-7171, an isopropyl ester
aryloxyphosphoramidate prodrug of PMPA (Scheme 137).
226
This method first involves
a DCC-mediated coupling between PMPA and phenol in NMP at high temperature.
The activation of the remaining hydroxyl with thionyl chloride and
the subsequent substitution with the amino acid isopropyl ester at
−78 °C afforded GS-7171 in 47% yield. At
this stage, R
p and S
p isomers were separated using simulated moving bed chromatography
to give GS-7340 (de > 98%).
Scheme 137
Syntheses of Both GS-7171 and GS-7340
Mackman et al. used a third strategy for the synthesis
of the phosphonomethyloxy
cyclic nucleoside GS-9131 (Scheme 138).
35
The conversion
of nucleoside 458 into phosphonate intermediate 461 was accomplished by oxidation
of the 5′-hydroxyl
using Jones’ reagent, glycal formation under Mitsunobu conditions,
and treatment with IBr and diphenyl hydroxymethyl phosphate. Oxidation
of the iodine with NaOCl and treatment with aqueous ammonia afforded
the nucleoside phenyl phosphonate monoester 462 in 18%
yield. Coupling of this compound with l-alanine ethyl ester
hydrochloride and PyBOP afforded the desired prodrug GS-9131 in 19% yield.
Scheme 138
Synthesis of GS-9131
3.3.2
3′,5′-Cyclic
Phosphoramidate
3′,5′-Cyclic phosphoramidates
have been recently
designed as an alternative to McGuigan Protides to mainly eliminate
the potential toxicity associated with the realease of phenol moieties.
Thus, Gardelli et al.
227
prepared 2′-C-methylcytidine-3′-5′-cyclic phosphoramidate 464 by reacting 2′-C-Me-C
with a chlorophosphoramidate
reagent bearing a 4-chlorophenol, in the presence of t-BuMgCl. At this stage, both
fast eluting (464a F.E.)
and slow eluting (464b S.E.) isomers were separated by
RP-HPLC and reacted with t-BuOK to form the desired
cyclic prodrugs. Interestingly, isomer 464a F.E. was
found to give the desired cyclic prodrug 465a (S
P) in 67% yield, while the other isomer 464b S.E. yields the corresponding cyclic
compound 465b
F.E. (R
P) in only 35% along with
monophosphate 466 (Scheme 139).
The absolute stereochemistry of the phosphorus center on both cyclic
compounds (S
P)-465a and (R
P)-465b was assigned by NOE experiments.
Scheme 139
2′-C-Me-Cytidine-3′-5′-cyclic
Phosphoramidate
Jain et al.
228
developed a one-step
method for the synthesis of 3′,5′-cyclic phosphoramidate
prodrug using novel phosphoramidate reagent 468 that
was prepared in two steps from 4-nitrophenol by first reaction with
POCl3 to give chloro intermediate 467, and
then reaction with alanine methyl ester in the presence of Et3N. Reaction of 5-fluoro-2′-deoxyuridine
(FdUrd) with
intermediate 468 in the presence of DBU afforded cyclic
prodrug 469 as a 5:1 mixture of diastereoisomers as determined
by 31P NMR (Scheme 140).
Scheme 140
2′-C-Me-FdU 3′-5′-Cyclic Phosphoramidate
3.3.3
Amino
Acid Amidate Monoester
Amino
acid phosphoramidate nucleoside monoester prodrugs were pioneered
in the 1990s by Wagner and co-workers.
229
This prodrug was designed as a modification of the aryloxyphosphoramidate
strategy detailed previously. The intention was to explore whether
the lipophilic aryl group was indispensable or not while increasing
the water solubility of the prodrug and losing the chirality of its
phosphorus center. It was designed in such a way that the amino acid
phosphoramidate mono ester biodegradation involves the direct cleavage
of the amino acid group by the action of a phosphoramidase (Figure 39).
144b,144c,230,231
Figure 39
Decomposition pathway
for amino acid amidate monoester nucleosides
prodrugs.
Interestingly, amino
acid phosphoramidate nucleoside monoesters
regained some interest in recent years with the discovery, by Herdewijn
et al., of their ability to act as a triphosphate mimic and thus to
be substrates of reverse transcriptases (including HIV-1). It was
demonstrated that amino acid phosphoramidate monoester nucleosides
with specific amino acids such as l-aspartic acid and particularly l-histidine, in
there acid form, are fulfilling the requirements
of structural and electronic properties that allow proper alignment
of α-phosphorus atom in the polymerase active site, mimicking
a nucleoside triphosphate (Figure 40).
232,233
The success of this recent approach led to its extension to modified
or unnatural amino acids derivatives.
234
Figure 40
Histidine phosphoramidate nucleoside monoester acting as a triphosphate
mimic.
The synthesis of amino acid phosphoramidate
mono ester nucleosides
prodrugs can be achieved by different synthetic pathways shown in
Figure 41: (A) formation of a hydroxyl cyanoethyl
nucleoside phosphite, oxidation to the monophosphate, and subsequent
coupling/deprotection, (B) formation of a methyl cyanoethyl nucleoside
phosphite and subsequent oxidative amination/deprotection, (C) formation
of a protected H-phosphonate, and subsequent oxidative
amination/deprotection, (D) hydrolysis of phosphorothioates, (E) hydrolysis
of phosphoramidates, (F) transformation of the nucleoside di- or triphosphate
by transient persilylation and substitution of a mono or diphosphate
unit, and (G) direct coupling of an amino acid with a nucleoside monophosphate.
Figure 41
Amino
acid phosphoramidate mono ester nucleoside formation.
Their phosphonate counterparts can be prepared
by coupling one
amino acid to phosphonate nucleoside (Figure 42).
Figure 42
Amino acid phosphonate mono ester nucleoside formation.
3.3.3.1
Amino Acid Phosphoramidate
Monoester
In 1994, the first synthesis of amino acid nucleoside
phosphoramidate
monoester was reported by Wagner and co-workers
235
to synthesize new AZT, FLT, and d4T prodrugs. AZT was first
reacted with 2-cyanoethyl-N,N-diisopropylchlorophosphoramidite
and subsequently treated with 1H-tetrazole and methanol
to yield the methyl cyanoethyl nucleoside phosphite intermediate 470. Subsequent reaction
with phenyl alanine methyl ester
in the presence of iodine and final hydrolysis afforded nucleoside
phosphoramidate 471 in 38% yield. Removal of the cyanoethyl
group with ammonia in methanol and purification on acidic resin afforded
the desired prodrug 472 in 88% yield (Scheme 141).
Scheme 141
First Synthesis of Amino Acid Nucleoside
Phosphoramidate Monoester
In 1996, a more commonly used method was reported by Abraham
et
al.
230a
for the synthesis of amino acid
phosphoramidate mono ester prodrugs of Ara-C and 5-FdU and involves
the direct coupling of the amino acid to the nucleoside monophosphate.
Thus, Ara-C was selectively phosphorylated at the 5′-position
with phosphorus oxychloride in triethyl phosphate. After hydrolysis,
a DCC-mediated coupling with phenylalanine and tryptophan methyl esters
afforded the desired amino acid Ara-C phosphoramidates 474a and 474b (Scheme 142).
Scheme 142
Ara-C
Monoester Phosphoramidates
In 1997, the same approach was used by Abraham et al.
for the preparation
of amino acid phosphoramidate monoester of acyclovir (Scheme 143).
236
After phosphorylation
of acyclovir, a DCC-mediated coupling with various amino esters afforded
the desired prodrugs 475 in 28–89% yield. It is
noteworthy that the coupling of ACV with cyanoethyl phosphate afforded
a mixture of products and was abandoned.
Scheme 143
Amino Acid Phosphoramidate
Monoester of Acyclovir
EDC has also been used as a coupling agent with 2′-deoxyadenosine
to perform the reaction at room temperature, and compound 476 was obtained in an improved
63% yield (Scheme 144).
230,234d
Scheme 144
EDC for Milder
Coupling Conditions
Interestingly, Abraham et al.
237
observed
a lack of selectivity between 5-FdU’s 5′- and 3′-hydroxy
groups during phosphorylation, and thus had to use a series of temporary
protecting groups. First, an MMtr group was introduced at the 5′-position
of 5-FdU before acetylation of the 3′-hydroxy group. Selective
5′-deprotection and DCC-mediated coupling with cyanoethyl phosphate
afforded protected 5-FdU intermediate 477. Deprotection
under basic conditions followed by coupling with phenylalanine or
tryptophan methyl esters afforded the desired 5-FdU prodrugs 479 (Scheme 145).
Scheme 145
5-FdU Prodrugs
Adelfinskaya et al. prepared
different amino acid 2′-deoxy
adenosine phosphoramidate mono esters (Scheme 146).
232
DCC-mediated coupling between 2′-deoxy
adenosine monophosphate 480 and various protected amino
acids afforded the amino acid methyl ester nucleoside phosphoramidates 481 in 39–94%
yield. The subsequent saponification
was carried out with 0.4 M NaOH to afford the amino acid nucleoside
phosphoramidates 482 in 22–94% yield. Alternative
deprotection with potassium carbonate in 2:1 MeOH:water could also
be used.
234a
A similar procedure was used
for the synthesis of 1-deaza-, 3-deaza-, and 7-deaza- adenine analogs
(not shown).
238
Scheme 146
Preparation of
Amino Acid 2′-Deoxy Adenosine Phosphoramidate
Mono Esters
Nucleoside phosphoramidate
monoesters bearing amino acid in their
acidic form, such as 484 and 486, are also
intermediates of aryloxyphosphoramidate bioactivation and were synthesized
as part of the activation studies (Scheme 147). These compounds were prepared by simple
saponification of their
corresponding aryloxyphosphoramidates 483 and 485, in aqueous triethylamine.
184b,201
Scheme 147
Synthesis of Phosphoramidate
Mono Acids 484 and 486
Alternatively, the amino acid nucleoside phosphoramidate
monoesters
can be generated using H-phosphonate intermediates.
239
AZT triethylammonium H-phosphonate
was generated in 69% yield from AZT by first treatment with diphenyl
phosphite in pyridine and subsequent hydrolysis in aqueous triethylamine.
Transient silylation of 487 followed by oxidation with
iodine, substitution with amino acids methyl ester, and basic hydrolysis
afforded the desired amino acid nucleoside phosphoramidate 488 in 31–70% yield. Treatment
with a methylamine solution in
methanol gave the corresponding methyl amide analogs 489 in good to quantitative yields
(Scheme 148).
Scheme 148
Amino Acid Nucleoside Phosphoramidate Monoesters, Generated
Using H-Phosphonate Intermediates
Other alkyl amines derivatives were directly
prepared from the H-phosphonate nucleoside as previously
described for the
methyl ester analogs in 28–56% yield (Scheme 149).
Scheme 149
Preparation of Alkyl Amine Derivatives from H-Phosphonate
Nucleosides
The same method
was used by Chang et al. for the preparation of
amino acid 2′,3′-dideoxyadenosine (ddA) phosphoramidate
monoester without requiring nucleoside protection.
240
The oxidative amination of the nucleoside H-phosphonate with different amino acids
afforded the desired ddA
prodrugs in yields ranging from 28% to 51% (Scheme 150).
Scheme 150
Preparation of Amino Acid ddA Phosphoramidate Monoester
Prodrugs
One major drawback
of such strategies is the need to use reverse
phase or ion-exchange chromatography for the purification of these
very polar nucleoside phosphate monoesters. To overcome this limitation,
Zhu et al.
241
used a fluorenylmethyl protecting
group (Scheme 151). A slight excess of diphenyl
phosphite was reacted successively with FmOH and with adenosine in
pyridine to afford nucleoside H-phosphonate intermediate 493. The Atherton–Todd amination
with amino acids hydrochloride,
carbon tetrachloride, and triethylamine afforded the protected nucleoside
phosphoramidates 494 in 73–82% yield. Deprotection
with piperidine in dichloromethane led to the desired amino acid adenosine
phosphoramidate mono esters 495 in 65–75% yields.
Ora et al. used a similar strategy to prepare l-alanine thymidine
phosphoramidate monoester (not shown).
212
Scheme 151
Fluorenylmethyl Protecting Group To Ease the Purification of
Highly
Polar Nucleoside Phosphate Monoesters
A benzyl group was also used as a temporary protecting
group by
Gardelli et al. for the preparation of amino acids 2′-C-Me-C phosphoramidate prodrugs.
242
Diphenyl phosphite was successively treated with the 2′-C-Me-C nucleoside and benzyl
alcohol to form nucleoside H-phosphonate 496 (Scheme 152). Oxidative amination with
l-alanine 2-propenylpentyl
ester under Atherton–Todd conditions afforded nucleosides 497. Subsequent acidic deprotection
with 80% TFA in water
and hydrogenolysis yielded the desired prodrugs 498 (yields
not provided).
Scheme 152
Benzyl Protecting Group To Ease the Purification of
Highly Polar
Nucleoside Phosphate Monoesters
Phosphite intermediates can also be generated through
phosphoramidites
as reported by Whalen et al. for the preparation of cytidine phosphoramidates
monoester.
243
Tribenzoyl cytidine phosphoramidite 501 was prepared by a tetrazole-mediated coupling
of tribenzoyl
cytidine with allyl phosphoramidite 500. Hydrolysis and
oxidative amination under Atherton–Todd conditions afforded
the protected nucleoside phosphoramidates 502 in 30–78%
yield. Removal of the allyl group with Pd(PPh3)4 and subsequent treatment with NaOMe
and NaOH afforded the desired
phosphoramidates 503 in 80% yield for all examples (Scheme 153).
Scheme 153
Use of Phosphoramidites To Generate Phosphoramidate
Prodrugs
Fu et al. reported
the synthesis of amino acid nucleoside phosphoramidate
monoester from nucleosides di- and triphosphates (Scheme 154).
244
Treatment of
ADP with TMSCl and various amino acid methyl ester in pyridine and
subsequent hydrolysis with 2 M ammonium hydroxide afforded the desired
nucleoside monophosphate prodrugs in roughly 50% yield.
Scheme 154
Synthesis
of Amino Acid Nucleoside Phosphoramidate Monoester from
Nucleosides Di- and Triphosphates
Using a similar approach, Zhu et al. prepared various
amino acid
nucleoside phosphoramidate monoesters from thymidine, uridine, adenosine,
and guanosine triphosphates (Scheme 155).
245
Scheme 155
Amino Acid Nucleoside Phosphoramidate
Monoesters from T, U, A, and
G Triphosphates
Amino acid nucleoside
phosphoramidate monoesters can also be obtained
by hydrolysis of phosphorothioamidate derivatives (Scheme 156).
216,246
2-Chloro-3-methyl-1,3,2-thiazaphospholidin-4-one
2-oxide was first reacted with glycine methyl ester hydrochloride
in the presence of triethylamine to provide intermediate 506. Subsequent reaction
with FLT and hydrolysis with 10% Et3N in dichloromethane over silica gel at 40 °C
afforded the amino
acid nucleoside phosphoramidate monoesters 508.
Scheme 156
Hydrolysis
of Phosphorothioamidates
De Napoli et al. used thymidine as a model for the synthesis
of
nucleoside phosphoramidates monoester libraries on solid phase.
247
Tentagel HL resin was first linked to the thymidine
phosphoramidite 509 via a 3-chloro-4-hydroxyphenylacetic
linkage by a tetrazole-mediated coupling. After oxidation with iodine
in pyridine and water, the 3′-trityloxy group was replaced
by an acetoxy group, and the cyanoethyl group was cleaved in the presence
of triethylamine in pyridine. With key intermediate 511 in hand, introduction of various
amino acids was carried out successfully
after tosylation of the phosphate ester. For optimal results, the
coupling was repeated three times before the desired nucleoside phosphoramidate
was detached from the solid support with concentrated aqueous ammonia
(Scheme 157).
Scheme 157
Synthesis of Nucleoside
Phosphoramidates Monoester Libraries on Solid
Phase
3.3.3.2
Amino Acid Phosphonamidate Monoester
The synthesis
of amino acid nucleoside phosphonamidate monoester,
such as 513, is much less represented in the literature
when compared to their phosphoramidate counterparts. McKenna et al.
248
reported the coupling of cidofovir with valine
methyl and ethyl esters in the presence of EDC in water in 35–40%
(Scheme 158).
Scheme 158
Synthesis of Cidofovir
Phosphonamidate Monoester Prodrug
A similar procedure was used by Adelfinskaya et al. for
the synthesis
of an aspartic acid adefovir derivative.
232b
DCC-mediated coupling of aspartic acid and adefovir and subsequent
saponification with sodium hydroxide in methanol and water afforded
the desired adefovir prodrug 514 in 58% yield (Scheme 159).
Scheme 159
Phosphonamidate of Adefovir
3.3.4
Borch’s
Methylaryl Haloalkylamidates
As part of their research program
on nucleosides, the Borch team
developed a neutral methyl aryl haloalkyl phosphoramidate prodrug
capable of passing through the cell membrane. These prodrugs are designed
to undergo intracellular activation to generate unstable phosphoramidate
anion intermediate B, which in turn undergoes spontaneous
cyclization and P–N bond cleavage by water to liberate the
nucleoside monophosphate (Figure 43). It is
noteworthy that their first prodrug approach involved a haloethyl
(instead of halobutyl) nucleoside phosphoramidate.
249
However, further mechanistic studies revealed that after
cyclization of the haloethyl phosphoramidate anion, nonselective nucleophilic
attack of water at the carbon and phosphorus centers of the pyrolidinium
ion intermediate was observed, delivering the NuMP, but also an undesired
hydroxyethyl byproduct.
250
Figure 43
Decomposition pathway
of Borch’s methylaryl haloalkylamidates
prodrugs.
The coupling of the
methyl aryl haloalkyl phosphoramidate moiety
to the nucleosidic part was achieved either using either P(III) or
P(V) chemistry (Figure 44). No chiral synthesis
has been developed so far, and final methylaryl haloalkyl phosphoramidate
prodrugs are always obtained as a mixture of diastereoisomers at the
phosphorus center.
Figure 44
P(III) versus P(V) chemistry.
This prodrug approach has been successfully applied to the
intracellular
delivery of anticancer nucleotide 5-fluoro-2′-deoxyuridine-5′-monophosphate
(FdUMP). This compound was synthesized using P(III) chemistry as shown
in Scheme 160.
251
Phosphorus trichloride is reacted with the corresponding alcohol
in the presence of diisopropylethylamine followed by reaction with N-methyl-N-(4-chlorobutyl)amine
hydrochloride
to generate chlorophosphoramidite 515. This intermediate
is directly reacted with 5FdU in situ and then oxidized with tert-butyl hydroperoxide
to yield 5-nitrofurfuryl N-methyl-N-(4-chlorobutyl) phosphoramidate 517 in 34% yield.
Scheme 160
FdU Borch’s Phosphoramidate
Wu et al.
252
investigated the influence
of hydrophilic modification of 5-FdU phosphoramidates by replacing
the N-methyl group with an N-dihydroxypropyl
chain. Selective phosphorylation on the 5′-hydroxyl group of
5-FdU using phosphoramidite 518, generated in situ by
reaction of N-allyl-4-chlorobutan-1-amine hydrochloride
with POCl3, provided compound 520. The -OBt
moiety was then displaced by either benzyl alcohol or 5-nitrofurfuryl
alcohol in the presence of DMAP to furnish the corresponding methylaryl
haloalkyl phosphoramidates derivatives. Dihydroxylation with OsO4/NMO afforded the
final N-2,3-dihydroxypropyl-N-(4-chlorobutyl) phosphoramidates 521 (Scheme 161).
Scheme 161
N-Dihydroxypropyl Phosphoramidates
Interestingly, attempts to
synthesize directly the methylaryl haloalkyl
phosphoramidate of cytosine derivatives, such as cytarabine
253
or gemcitabine,
254
using either P(III) or P(V) approaches, were unsuccessful presumably
because of possible side reactions and very low solubility of the
nucleosides. To circumvent this problem, the cytosine amino group
had to be protected with an allyloxycarbonyl group that was removed,
after the phosphorylation step, by treatment with Pd(PPh3)4 and p-toluenesulfinate
(Scheme 162).
Scheme 162
Allyloxycarbonyl Group as Transient Protective
Group
3.4
Phosphorodiamidates and Phosphonodiamidates O-PO(NR2)2 and C-PO(NR2)2
Phosphorodiamidate prodrugs have rarely
been used for the last 20 years, probably due to the success of aryloxyphosphoramidates.
It has only been recently that this prodrug approach was reinvestigated
because, unlike aryloxyphosphoramidates, it bears an achiral phosphorus
center and releases only natural amino acids upon metabolism.
A putative mechanism of unmasking to the monophosphate was proposed
by McGuigan et al.
255
in which carboxypeptidase
cleaves the ester function of the amino acid, inducing spontaneous
cyclization of the carboxylate of the free amino acid onto the phosph(on)ate
moiety. After a spontaneous hydrolysis, the nucleoside phosphoramidate
monoester is cleaved into the free nucleoside monophosph(on)ate by
action of phosphoramidase (Figure 45).
Figure 45
Mechanism
of action of O- and C-phosphorodiamidate
nucleoside prodrugs.
Bis(amino acid) nucleoside phosphorodiamidates can be prepared
as shown in Figure 46: (A) phosphorylation
of the nucleoside with phosphorus oxychloride and subsequent bis-substitution
with amino acids, and (B) chlorination of a nucleoside monophosphate
and coupling with the amino acids.
Figure 46
Methods to access O-phosphorodiamidate
nucleoside
prodrugs.
On the other hand,
the synthesis of the bis(amino acid) nucleoside C-phosphorodiamidate involves three
different methods highlighted
in Figure 47: (A) direct coupling of the phosphonic
acid nucleoside with amino acids, (B) TMSBr-deprotection of the phosphonate
alkyl ester nucleoside and subsequent coupling with amino acids, and
(C) chlorination of a nucleoside phosphonate and coupling with amino
acids.
Figure 47
Methods to access C-phosphorodiamidate nucleoside
prodrugs.
3.4.1
Bis(amino
acid) O-Phosphorodiamidates
The first syntheses
of bis(amino acid) O-phosphorodiamidate
nucleosides were described in 1991 by McGuigan and co-workers who
used AZT
256
and FdU
257
as substrates. AZT was reacted with phosphorus oxychloride
in triethylphosphate to generate AZT monophorodichloridate 525. Subsequent substitution
with excess amino acids in the presence
of triethylamine afforded the corresponding AZT phosphorodiamidate
prodrugs 526 in 21–44% yield (Scheme 163).
Scheme 163
AZT O-Phosphorodiamidates
Synthesis
McGuigan et al.
also reported the preparation of bis(amino acid)-2′-methyl-6-methoxyguanosine O-phosphorodiamidate
prodrug as part of an extensive SAR
study (Scheme 164).
255
Compounds were prepared either using the conditions described above
or by slight modification of the procedure. Nucleoside was first phosphorylated
with POCl3 at −78 °C in THF. Subsequent displacement
with amino acids was carried out in the presence of diisopropylethylamine.
Scheme 164
2’-Methyl-6-alkoxyguanosine Phosphorodiamidate Prodrug
AA = amino acid.
Nonsymetrical O-phosphorodiamidates have
been
obtained in yields ranging from 4% to 17%
255
following a similar protocol with successive addition of two different
amino acids (Scheme 165).
Scheme 165
Nonsymetrical O-Phosphorodiamidates
Key phosphorodichloridate intermediates can be alternatively
generated
from a nucleoside monophosphate as reported by Korboukh et al.
258
Nucleoside monophosphate 532 was
obtained in three steps through phosphoramidite coupling, subsequent
oxidation, and cleavage of the tert-butyl groups
under acidic conditions. Compound 532 was then reacted
with 3 equiv of oxalyl chloride and a catalytic amount of DMF to form
phosphorodichloridate intermediate 533. Subsequent reaction
with 2.5 equiv of ethyl glycine hydrochloride in the presence of DIPEA,
and isopropylidene deprotection with para-toluene
sulfonic acid in methanol at 60 °C afforded desired prodrug 534 in 7% yield (Scheme
166).
Scheme 166
O-Phosphorodiamidates Nucleoside Prodrugs from Phosphate
Nucleosides
3.4.2
Bis(amino
acid) C-Phosphorodiamidates
The bis(amino
acid) nucleoside phosphorodiamidates are generally
obtained from phosphonic acid intermediates after activation as phosphorodichloridates.
Thus, Serafinowska et al. reported the synthesis of acyclophosphonate
prodrug 538(259) in 15% yield
by treatment of ethyl phosphonate derivative 535 with
TMSBr, reaction of the corresponding silyl ester 536 with
PCl5, and reaction with alanine methyl ester hydrochloride
in the presence of triethylamine and NMI (Scheme 167).
Scheme 167
Synthesis of [(Phosphonomethoxy)ethoxy]adenine Prodrug 538
Formation of such
bis(amino acid) nucleoside phosphorodiamidates
can also sometimes require temporary protection of the nucleobase.
Thus, Dang et al. (Scheme 168, eq 1)
260
treated PMEA with oxalyl chloride in the presence
of DMF, allowing simultaneous chlorination of the phosphonic acid
and protection of the N
6-position. Subsequent
reaction with 2-methylalanine ethyl ester and triethylamine gave the
protected diamidates prodrugs 541. Finally, hydrolysis
of the formidamide protection with acetic acid in isopropanol afforded
the desired PMEA prodrug 542 in 16% yield. Interestingly,
reactions carried out with the glycine methyl ester without nucleobase
protection failed to produce the corresponding prodrug.
Bis(amino
acid) PMEA prodrugs such as 543(260) have also been prepared by direct coupling
of PMEA with glycine ethyl ester in pyridine and treatment with a
premixed solution of triphenylphosphine and 2,2′-dipyridyl
disulfide (Scheme 168, eq 2). Interestingly,
the reaction was reported to fail when using 2-methylalanine ethyl
ester presumably because of the steric hindrance at the nitrogen.
Scheme 168
Synthesis of PMEA Bis(amino acid) Nucleoside Phosphorodiamidates
with Protective Groups
A similar procedure was used by Mackman et al.
35
for the synthesis of GS-9148 bis(amino
acid)
prodrug derivative (Scheme 169). Diethyl nucleoside
phosphonate 544 was first treated with ammonium hydroxide
generating 6-aminopurine nucleobase while deprotecting one of the
phosphonate esters. Subsequent treatment with TMSBr afforded phosphonic
acid 545. The coupling of different amino acids in the
presence of 2,2′-dithiopyridine, triphenyl phosphine, and triethylamine
in pyridine afforded the desired prodrugs 546 in 11–73%
yield.
Scheme 169
GS-9148 Bis(amino acid) Prodrug
Jansa et al.
261
reported the synthesis
of bis(amino acid) nucleoside phosphonamidate prodrugs directly from
the bis(alkyl) nucleoside phosphonate 549 by coupling
of transient silyl ester phosphonate intermediate 548 with amino acids (Scheme 170).
This procedure
prevents tedious isolation of nucleoside phosphonic acids.
Scheme 170
Nucleoside
Phosphonamidate Prodrugs Directly from the Bis(alkyl)
Nucleoside Phosphonates
4
Nucleoside Di- and Triphosphate
Prodrugs
For two decades, numerous prodrug strategies have
been developed
to deliver nucleoside monophosphates into the cells. The monophosphate’s
delivery allows bypassing the first phosphorylation, which is often
the rate-limiting step to NTP formation. However, di- and triphosphate
prodrugs have rarely been studied. This lack of research can be explained
by the generally efficient second and third phosphorylations (for
most nucleosides) and the inherent instability of the phosphate anhydride
bond. This bond is only kinetically stable because of the negative
charge resonance that avoids the nucleophilic attack at phosphorus
moiety.
The di- and triphosphate prodrug strategy has been mainly
applied
to AZT. AZT is a highly potent anti-HIV drug that was the first FDA-approved
nucleoside analog for treatment of HIV infection. AZT is efficiently
converted to the monophosphate, but only slowly to the diphosphate,
262
resulting in intracellular accumulation of
AZT-MP, which is responsible for some of its side effects.
263
In an ideal situation, the delivery of AZT-DP
or AZT-TP would retain the antiviral activity of the parent compound,
but avoid the toxicity associated with the AZT-MP. This approach was
also widely applied to ara-C to increase its bioavailability and to
avoid base deamination.
The main strategy for the synthesis
of di- and triphosphate prodrugs
developed in the early 1980s involves the introduction of an alkyl
or acyl lipophilic chain to the last phosphate unit (β-phosphorus
for diphosphate and γ- for triphosphate). The synthesis of these
lipophilic di- and triphosphate prodrugs commonly involves the coupling
of a lipophilic chain bearing a phosphate or pyrophosphate moiety
to a NDP or NTP. The lipophilic chain itself can be also directly
coupled to a NDP or NTP. Recently, a new strategy was developed as
acyloxybenzyl β-diester diphosphate using P(III) chemistry with
the coupling of a phosphoramidite and a NuMP (Figure 48).
Figure 48
BLG = biolabile group.
4.1
Nucleoside Di- and Triphosphate Glycerides
Several nucleoside di- and triphosphate prodrugs bearing acyl and
alkyl glyceride moieties have been reported. The rational design of
these prodrugs was based on naturally occurring phospholipid cytidine
diphosphate diglyceride, which is a natural intermediate in the biosynthesis
of anionic glycerophospholipid in mammalian cells. These prodrugs
were mainly developed to reach HIV reservoirs such as macrophages
and related cells involved in phagocytosis and antigen presentation.
However, administration of antiviral nucleosides such as AZT, ddC,
or 3′-deoxythymidine as nucleoside diphosphate diglycerides
was found to deliver monophosphorylated anti-HIV agents intracellularly,
due to the cleavage of the pyrophosphate unit between the α-
and β-phosphorus (Figure 49).
264
Figure 49
Mechanism of action for nucleoside diphosphate
glycerides.
Phosphatidic acid was
commonly used for the synthesis of nucleosides
di- and triphosphate diglycerides. Historically, the direct coupling
of nonactivated phosphatidic acid and nucleoside monophosphate was
first reported on natural nucleosides but provided only low yields.
265
Therefore, the most common strategy involves
the coupling of phosphatidic acid to a nucleoside 5′-monophosphate
activated as a morpholidate (Scheme 171).
266,267
The activation of the phosphatidic acid rather than the nucleoside
monophosphate was reported later to give better yields and to facilitate
the purification step.
268
NTPs diglycerides
were less described and generally synthesized by coupling of phosphatidic
acid and an activated NDP in low yield: for instance, AZT-TP distearoylglycerol
was prepared by condensation of AZT diphosphate with distearoylphosphatidic
acid morpholidate. It was shown to deliver a mixture of AZT and AZT-MP
to the cells.
269
Numerous saturated and
unsaturated lipophilic chains linked to the glycerol moiety have been
reported such as myristyl, palmityl, stearyl, or oleyl.
The
coupling of a morpholidate activated phosphatidic acid was
also used for the preparation of a myristoyl glyceride DP derivative
of acyclovir.
268b
This compound (not shown)
was found to be active on ACV resistant herpes TK–, indicating an efficient delivery
of ACV-MP.
270
This prodrug approach was also applied to vidarabine
(ara-A) and
cytarabine (ara-C), which are known for their antiviral and anticancer
activity, respectively (not shown). These compounds bearing free hydroxyl
groups in 2′- and 3′-position did not require any protection
to perform the synthesis of their diphosphate prodrugs. While NDP-prodrugs
were found less active that parent ara-C in in vitro antiproliferative
studies, they were actually much more potent in mice.
271
In the form of a diphosphate prodrug, ara-C
was found to be protected from the cytosine deamination, which leads
to the biologically ineffective ara-U.
272
Scheme 171
AZT DP-Prodrug
Some studies also reported the synthesis of oxyalkyl and
thioalkyl
ether glyceride of anti-HIV
273
and anticancer
274
agents. The synthetic strategy remains the
same with the coupling of the glycerophospholipid part to a NMP activated
as morpholidate (Scheme 172). The thioalkyl
and oxyalkyl ether glycerophospholipid were previously synthesized
by successive alkylation and acylation. After removal of the protective
trityl group, the alcohol is treated with POCl3 followed
by hydrolysis.
Scheme 172
Synthesis of Oxyalkyl and Thioalkyl Ether Glycerides
Ara-C-DP Prodrugs
4.2
Lipids and Steroids Nucleoside Di- and Triphosphates
or Phosphonophosphates
4.2.1
Acyl Phosphates
The literature
reports several examples of lipophilic acyl chains linked to the NDP
or NTP. After cell penetration, the acylphosphate is expected to be
cleaved by a hydrolase to give the corresponding NDP or NTP (Figure 50). Interestingly,
the preferential cleavage of
the mixed carboxylic phosphoric anhydride part (C–O–P)
over the phosphoric anhydride (P–O–P) was observed in
buffer and in culture media. Thus, the instability of this prodrug
in cell culture media did not allow for an efficient transmembrane
diffusion resulting in poor cellular uptake.
275
Figure 50
Expected and observed mechanisms of acyl phosphate nucleoside prodrugs.
The syntheses of octanoyl, lauroyl,
myristoyl, and palmitoyl acyl
nucleoside diphosphates of AZT (not shown) and d4T (560) were first reported by coupling
an acyl pyrophosphate unit to a
nucleoside with DCC. It is noteworthy that better yields were observed
when the tetrabutylammonium counterions of the acyl pyrophosphate
were exchanged for tributylammonium. Acyl nucleoside triphosphates
(561) on the other hand were obtained by coupling of
a phosphoro morpholidate nucleoside and an acyl pyrophosphate (Scheme 173).
276
Scheme 173
Synthesis
of d4T Octanoyl, Lauroyl, Myristoyl, and Palmitoyl Acyl
Nucleoside Diphosphates and Triphosphates
An alternative procedure for direct DCC-coupling of an
acyl chain
to the NDP or NTP was developed by Kreimeyer et al.
277
This method appeared to be efficient for the formation
of 2′,3′-deoxynucleosides prodrugs, but low yields were
observed with ribofuranosyl purine nucleotides due to additional potential
acylation sites. To circumvent this problem, the authors used an ethyl
chloroformate activated form of myristoic acid that selectively reacted
with ADP or ATP in good yields (Scheme 174).
Scheme 174
ADP or ATP Prodrugs
Kreimeyer et al.
278
used a
similar approach
to prepare a cholesterol carbonate prodrug of adenosine triphosphate 563 and showed
that this compound was effectively transported
across the membrane bilayer of liposomes (Figure 51).
Figure 51
Cholesterol carbonate prodrug of ATP 563.
4.2.2
Ether
Phosphates
Steroids and lipids
ether diphosphates nucleosides were also developed by Hong et al.
Because of the ether linkage between the lipid chain and the phosphorus
moiety, hydrolysis by hydrolase is impossible, but the intracellular
cleavage of the pyrophosphate unit allows for the delivery of nucleoside
monophosphate (Figure 52).
279
Figure 52
Mechanism of action of ether phosphates.
Steroids diphosphate derivatives of ara-C were
prepared by coupling
between ara-CMP morpholidate and various phosphocorticosteroids (Scheme 175). These
phosphocorticosteroids were synthesized
by either condensation of the steroid 564 with 2-cyanoethylphosphate
in the presence of DCC followed by deprotection of the cyanoethyl
group or by treatment of 21-iodocorticosteroid 565 with
phosphoric acid. This second method was generally preferred because
of easy purification of product 566 by simple crystallization.
It is noteworthy that these corticosteroid diphosphate prodrugs of
ara-C, 567, showed similar activities in vitro as compared
to their corresponding monophosphate prodrugs, but were found to be
generally less active in vivo. These differences were attributed to
the high hydrolysis rate of the phosphoric anhydride bond of the diphosphate
prodrugs.
Scheme 175
Preparation of Ara-C Steroids Diphosphate Derivatives
4.2.3
Phosphonophosphates
Derivatives
Alkyldiphosphate and alkylphosphonophosphate
derivatives of naturally
occurring nucleosides such as cytidine, deoxycytidine, thymidine,
and adenosine have been reported to exhibit antiproliferative activities
that were attributed to the phospholipidic chain.
280,281
Thus, based on this work, alkylphosphono phosphate ara-C derivatives,
(compound 568 is shown in Scheme 176 as a representative example) were prepared as
a prodrug that would
increase the bioavailability of the nucleoside while avoiding the
deamination of the cytosine occurring at the nucleoside level and
deliver ara-CTP and a phospholipidic chain, two cytotoxic principles.
282
Scheme 176
Preparation of ara-C Alkylphosphono Phosphate
Derivatives
Ruiz et al.
283
reported the synthesis
of PMEA and HPMPC phosphonophosphate HDP and ODE prodrugs (Scheme 177). The phosphate
bearing the lipophilic group
was obtained by reaction of phosphorus oxychloride and HDP–OH
or ODE–OH. Alkoxyalkylphosphates were then activated as phosphomorpholidates
using DCC, followed by reaction with DMTr-protected HPMPC in the presence
of pyridine. Following DMT deprotection with TFA, phosphonophosphates
HDP and ODE prodrugs were afforded in 40% and 20% yield, respectively.
Unfortunately, these compounds were found to exert less antiviral
activity than their HDP and ODE phosphonate prodrugs.
Scheme 177
Synthesis
of PMEA and HPMPC Phosphonophosphate HDP and ODE Prodrugs
4.3
para-Methoxybenzyl Diphosphate
Diester
More recently, Meier’s group proposed to use
various biolabile protecting groups to synthesize diphosphate prodrugs
to efficiently deliver diphosphate nucleosides.
284
The first attempt was realized using a cycloSal (see section 3.1.3) protecting group
to
mask both hydroxyl groups of the β-phosphate moiety. However,
after preparation of several aryl substituted cycloSal diphosphate (synthesis not
reported), they observed the predominant
release of NMP, by hydrolysis of the phosphorus anhydride bond. To
circumvent the hydrolysis of the pyrophosphate unit, the use of a para-acyloxybenzyl
(see section 3.1.3) protecting group was investigated (Figure 53). Unlike the cycloSal,
deprotection of para-acyloxybenzyl is initiated by enzymatic or chemical
cleavage of the ester group, and not by nucleophilic attack at the
phosphorus moiety.
Figure 53
Use of a para-acyloxybenzyl group to
synthesize
diphosphate prodrugs.
AZT (not shown) and d4T (574) DP prodrugs were
synthesized
using P(III) chemistry via a dicyanoimidazole-mediated coupling of
bis-para-acyloxybenzylphosphoramidite, 573, and bis(tetra-n-butylammonium)
nucleoside monophosphates, followed by a subsequent oxidation with tert-butyl hydroperoxide.
Phosphoramidites were previously
synthesized by reaction of para-acyloxybenzylalcohol
and diisopropyl phosphoramidous dichloride (Scheme 178). Interestingly, these compounds
proved to possess a high
chemical stability in buffer but also to undergo fast and highly selective
enzymatic cleavage in cell extract to deliver NDPs. The retained antiviral
activities of d4T diphosphate prodrugs (no marked toxicity) proved
their ability to penetrate the cells and release biologically active
metabolites intracellularly.
Scheme 178
AZT and d4T Bis(para-scyloxybenzyl)-phosphoramidites
DP Prodrugs
5
Conclusion
Despite that the concept of phosph(on)ates prodrugs
was originally
developed in the 1990s and led to the FDA approval of potent antiviral
such as TDF in 2001, it is only very recently that the synthesis of
phosph(on)ate prodrugs became systematic in the nucleoside field.
Indeed, the large number of examples in the literature of phosph(on)ate
prodrugs increasing the activity of a nucleoside or even better, revealing
the activity of a inactive parent nucleoside, has led the nucleoside
community to consider prodrug evaluation as indispensable. As presented
in this Review, multiple synthetic methodologies were developed to
prepare a large variety of phosph(on)ates prodrugs. However, several
challenges remain, including the development of efficient methods
for the preparation of chiral phosph(on)ates prodrugs because one
diastereomer may possess overall biological properties superior to
those of the other. Improvements in the targeting of prodrugs to particular
organs and cellular compartment as well as the development of nanoparticles
containing nucleoside prodrugs are needed. Finally, the ultimate goal
remains the development of efficient triphosphate prodrugs that would
completely overcome the phosphorylation issues by delivering the active
compound directly to the target polymerase. The application of prodrug
technology has had a large impact on the development of nucleoside
and nucleotide antiviral therapies, and provides great hope for persons
suffering from deadly viruses such as HIV, HBV, and HCV. Finally,
it is likely that the lessons learned from these viruses with novel
nucleoside prodrugs will be applied to new emerging viruses such as
Noro, Hendra, Dengue, and Chikungunya viruses.
Related collections
Most cited references83
- Record: found
- Abstract: found
- Article: not found
Adefovir dipivoxil for the treatment of hepatitis B e antigen-positive chronic hepatitis B.
Lennox Jeffers, M Shiffman, Terry Fry … (2003)
- Record: found
- Abstract: found
- Article: not found
Clinical potential of the acyclic nucleoside phosphonates cidofovir, adefovir, and tenofovir in treatment of DNA virus and retrovirus infections.
Erik De Clercq (2003)
- Record: found
- Abstract: not found
- Article: not found
Force Spectroscopy of Molecular Systems—Single Molecule Spectroscopy of Polymers and Biomolecules
Andreas Janshoff, Marcus Neitzert, York Oberdörfer … (2000)
