- Record: found
- Abstract: found
- Article: found
Exposure to dietary lipid leads to rapid production of cytosolic lipid droplets near the brush border membrane

Read this article at
Abstract
Background
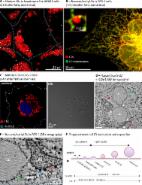
Intestinal absorption of dietary lipids involves their hydrolysis in the lumen of proximal intestine as well as uptake, intracellular transport and re-assembly of hydrolyzed lipids in enterocytes, leading to the formation and secretion of the lipoproteins chylomicrons and HDL. In this study, we examined the potential involvement of cytosolic lipid droplets (CLD) whose function in the process of lipid absorption is poorly understood.
Methods
Intestinal lipid absorption was studied in mouse after gavage. Three populations of CLD were purified by density ultracentrifugations, as well as the brush border membranes, which were analyzed by western-blots. Immunofluorescent localization of membranes transporters or metabolic enzymes, as well as kinetics of CLD production, were also studied in intestine or Caco-2 cells.
Results
We isolated three populations of CLD (ranging from 15 to 1000 nm) which showed differential expression of the major lipid transporters scavenger receptor BI (SR-BI), cluster of differentiation 36 (CD-36), Niemann Pick C-like 1 (NPC1L1), and the ATP-binding cassette transporters ABCG5/G8 but also caveolin 2 and fatty acid binding proteins. The enzyme monoacylglycerol acyltransferase 2 (MGAT2) was identified in the brush border membrane (BBM) in addition to the endoplasmic reticulum, suggesting local synthesis of triglycerides and CLD at both places.
Conclusions
We show a very fast production of CLD by enterocytes associated with a transfer of apical constituents as lipid transporters. Our findings suggest that following their uptake by enterocytes, lipids can be partially metabolized at the BBM and packaged into CLD for their transportation to the ER.
Related collections
Most cited references59
- Record: found
- Abstract: found
- Article: not found
Triacylglycerol synthesis enzymes mediate lipid droplet growth by relocalizing from the ER to lipid droplets.
- Record: found
- Abstract: found
- Article: not found
Proteomic analysis of proteins associated with lipid droplets of basal and lipolytically stimulated 3T3-L1 adipocytes.
- Record: found
- Abstract: found
- Article: found