- Record: found
- Abstract: found
- Article: found
Antiplatelet regimens in the long-term secondary prevention of transient ischaemic attack and ischaemic stroke: an updated network meta-analysis
Read this article at
Abstract
Objective
To examine the comparative efficacy and safety of different antiplatelet regimens in patients with prior non-cardioembolic ischaemic stroke or transient ischaemic attack.
Data sources
As on 31 March 2015, all randomised controlled trials that investigated the effects of antiplatelet agents in the long-term (≥3 months) secondary prevention of non-cardioembolic transient ischaemic attack or ischaemic stroke were searched and identified.
Outcome measures
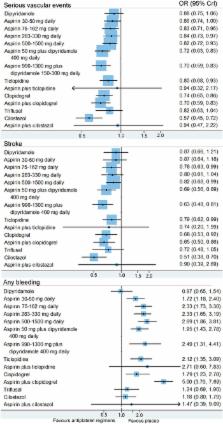
The primary outcome measure of efficacy was serious vascular events (non-fatal stroke, non-fatal myocardial infarction and vascular death). The outcome measure of safety was any bleeding.
Results
A total of 36 randomised controlled trials (82 144 patients) were included. Network meta-analysis showed that cilostazol was significantly more effective than clopidogrel (OR 0.77, 95% credible interval 0.60–0.98) and low-dose (75–162 mg daily) aspirin (0.69, 0.55–0.86) in the prevention of serious vascular events. Aspirin (50 mg daily) plus dipyridamole (400 mg daily) and clopidogrel reduced the risk of serious vascular events compared with low-dose aspirin; however, the difference was not statistically significant. Furthermore, low-dose aspirin was as effective as higher daily doses. Cilostazol was associated with a significantly lower bleeding risk than most of the other regimens. Moreover, aspirin plus clopidogrel was associated with significantly more haemorrhagic events than other regimens. Direct comparisons showed similar results as the network meta-analysis.
Conclusions
Cilostazol was significantly more effective than aspirin and clopidogrel alone in the long-term prevention of serious vascular events in patients with prior non-cardioembolic ischaemic stroke or transient ischaemic attack. Cilostazol was associated with a significantly lower bleeding risk than low-dose aspirin (75–162 mg daily) and aspirin (50 mg daily) plus dipyridamole (400 mg daily). Low-dose aspirin was as effective as higher daily doses. However, further large, randomised, controlled, head-to-head trials are needed, especially in non-Asian ethnic groups.
Related collections
Most cited references50
- Record: found
- Abstract: found
- Article: not found
Collaborative meta-analysis of randomised trials of antiplatelet therapy for prevention of death, myocardial infarction, and stroke in high risk patients.
- Record: found
- Abstract: found
- Article: not found
A randomised, blinded, trial of clopidogrel versus aspirin in patients at risk of ischaemic events (CAPRIE). CAPRIE Steering Committee.
- Record: found
- Abstract: found
- Article: not found