Introduction
Coronary artery disease (CAD) is one of the main causes of death and morbidity at a global level, and it has resulted in an enormous economic burden. Diabetes mellitus is an independent risk factor for CAD [1] and increases the risk of vascular outcomes such as ischemic stroke, CAD, and vascular death [2]. Diabetes mellitus often leads to secondary complications, such as nerve damage, kidney damage, and retinopathy [3]. The high prevalence, disability, and mortality of patients with diabetes mellitus have made it a serious health problem worldwide [4–6]. According to large-scale national research in China, 11.2% of adults (according to WHO criteria) or 12.8% of adults (ADA criteria including the addition of HbA1c) aged 18 and older living in China had diabetes mellitus in 2017, and the total number of affected patients was estimated to be 129.8 million (70.4 million men and 59.4 million women) [7]. Patients with type 2 diabetes mellitus (T2DM) are twice as likely to develop cardiovascular disease as patients without diabetes mellitus [8].
However, it has been suggested that patients with prediabetes may experience functional decline and disability before the onset of full-scale diabetes mellitus [9]. Specifically, blood glucose level variability in patients with prediabetes is significantly higher, even in the early stages of glucose metabolism disorder. Thus, blood glucose level variability is an additional parameter to assess glucose homeostasis [10] and it is also a condition that puts an individual at high risk of diabetes mellitus and its complications [11]. Since a large proportion of patients with prediabetes progress to T2DM, and the risk of vascular consequences caused by abnormal blood glucose levels therefore increases [12], early intervention and management is very important for the care of patients with prediabetes. According to the American Diabetes Association, glycosylated hemoglobin (HbA1c) is the basis for diagnosing prediabetes (5.7–6.4%, or 39–47 mmol/mol) [13]. Compared with measurement of fasting blood glucose (FBG) level and the oral glucose tolerance test, measurement of HbA1c level is more convenient as it does not require fasting, HbA1c level is stabler before analysis, and HbA1c level has less daily variation with stress, dietary changes, or illness [14]. There is a strong and persistent association between HbA1c level and subsequent progression to diabetes mellitus [15]; moreover, the predictive HbA1c level of 5.7–6.4% for diabetes mellitus progression is similar to that of FBG assessment alone [16]. A few studies have shown that HbA1c level is associated with the prognosis of patients with prediabetes. Therefore, we chose HbA1c level as the research index in this study. Although prediabetes can cause significant harm [17, 18], it has not yet attracted sufficient attention, and there have been few related studies. In addition, the prognostic value of HbA1c level in individuals with prediabetes and CAD is still unclear worldwide, and it has not been discussed in Asian populations although the reference HbA1c level is different among different races [19]. Therefore, we studied the relationship between HbA1c level and adverse cardiovascular outcomes in Asian patients with prediabetes and CAD.
Methods
Study Population
This was a single-center, retrospective, empirical observational study. We selected 1672 successive patients with prediabetes and CAD from Beijing Anzhen Hospital from August 2018 to January 2021. This study was approved by the Institutional Ethics Committee of Beijing Anzhen Hospital. Since the data were obtained from electronic medical records at the time of admission, written informed consent of the participants was not required. The inclusion criteria encompassed no history of diabetes mellitus, no history of taking hypoglycemic drugs or using insulin, and an HbA1c level of 5.7–6.4%. The exclusion criteria were as follows: (1) cardiogenic shock; (2) renal insufficiency or renal replacement therapy with an estimated glomerular filtration rate (eGFR) below 30 mL/(min·1.73 m2); (3) chronic infectious diseases; (4) history of malignant tumors; (5) percutaneous coronary intervention (PCI) failure and related complications, and hospital death; and (6) missing clinical data. Finally, this study included 1367 patients (Figure 1).
Data Collection and Definitions
We used standard case report forms to collect the demographic and clinical characteristics of each patient, including age; body mass index (BMI); blood pressure (systolic and diastolic blood pressure); smoking history and drinking history; medical history, including hypertension and hyperlipidemia; and medications. A venous blood sample was taken 1 day after the coronary procedure, and high-sensitivity C-reactive protein, triglyceride, cholesterol, low-density lipoprotein cholesterol (LDL-C), high-density lipoprotein cholesterol (HDL-C), creatine kinase, creatine kinase myocardial band, serum creatinine, uric acid, FBG, and HbA1c levels and levels of other biomarkers were analyzed at the Laboratory of Beijing Anzhen Hospital. On admission, blood pressure was measured twice with the patient in a calm state; hypertension was considered when individuals were taking antihypertensive drugs and/or blood pressure was above 140/90 mmHg. Dyslipidemia was diagnosed when fasting triglyceride level was above 150 mg/dL, and/or total cholesterol level was above 200 mg/dL, and/or LDL-C level was above 3.30 mmol/L, and/or HDL-C level was below 1.00 mmol/L, and/or when individuals were taking lipid-lowering drugs over a long period. BMI was defined as weight (kg) divided by height (m) squared. eGFR was calculated as follows: eGFR [mL/(min*1.73m2)] = 186*[serum creatinine (μmol/L)×0.011312]-1.154*age-0.203 (*0.742 if female) [20]. According to the HbA1c level on admission, we divided the patients into four groups: group 1 (n=444, HbA1c level ≤ 5.8%), group 2 (n=389, 5.9% ≤ HbA1c level ≤ 6.0%), group 3 (n=380, 6.1% ≤ HbA1c level ≤ 6.2%), and group 4 (n=167, HbA1c level ≥ 6.3%). The implementation of PCI was in line with China’s current clinical practice guidelines, and appropriate decisions were made by experienced interventional cardiologists [21].
Follow-up and End Point Events
After baseline coronary angiography, all of the patients were regularly followed up by a well-trained clinician who was blinded to the purpose of the study. They were followed up after 3, 6, and 12 months, and then each year until 43 months. Information about the outcomes was collected through telephone surveys of patients or their families. If necessary, this information was further verified in medical records. The primary end point was a complex of events including nonfatal myocardial infarction (MI), hospitalization for unstable angina, and ischemia-driven revascularization (IDR). The secondary end points were each of the elements of the complex primary end point. MI was defined as an increase in cardiac troponin level above the upper reference limit, and symptoms and/or ECG findings suggesting ischemia with or without ST-segment elevation. Hospitalization for unstable angina was defined as hospitalization for the treatment of unstable angina occurring within 24 hours from the latest symptoms. IDR was defined as the process of revascularization combined with changes in ECG and/or ischemic symptoms [22]. The primary end point event that occurred during the follow-up period was used in this research. In patients with various negative outcomes during the follow-up period, only the most serious events (nonfatal MI > readmission for unstable angina > IDR) were selected for the analysis. If the same type of event occurred more than once, only the first occurrence of the event was used for the analysis. The events were followed up until January 2021.
Statistical Analysis
Continuous variables were represented as the mean ± standard deviation, and the difference between the two groups was evaluated with the independent-sample t test or the Mann-Whitney U test, depending on the normality of the distribution. Categorical variables were described as numbers and percentages, and the chi-square test or Fisher’s exact test was used to analyze intergroup differences. The Spearman rank correlation test or the Pearson correlation test was used to evaluate the relationship between HbA1c level and cardiovascular adverse outcomes. Specifically, the Pearson correlation test was used to determine the association between two continuous variables that satisfied a normal distribution; in contrast, if one or more variables were not normally distributed and for categorical variables, we used the Spearman rank correlation test. The Kaplan-Meier method was used to calculate the event rate during the follow-up and to plot the time-event curve. A Cox proportional-hazards regression analysis was conducted to determine the hazard ratio (HR) with 95% confidence intervals (CIs). HbA1c level was used both as a continuous variable and as a categorical variable. Factors associated with the development of primary end points identified by univariate analysis were then examined by multivariate analysis. In the multivariate model, the following confounding factors were selected on the basis of either their clinical relevance or the results of the univariate analysis: age, male sex, BMI, previous MI, smoking status, drinking status, hypertension, dyslipidemia, uric acid level, stent implantation, use of a drug-coated balloon, and number of stents. We used IBM SPSS Statistics 23.0 (IBM, Armonk, NY, USA) for statistical testing. The level of significance was set at P<0.05.
Results
The age of the 1367 patients at the baseline was 58 ± 10.3 years, and 71.6% of patients were men (n=979). During 43 months of follow-up, 197 patients (14.4%) experienced a primary end point event, including 37 individuals with nonfatal MI, 189 with hospitalization for unstable angina, and 90 with IDR.
Baseline Characteristics of the Study Population
The baseline characteristics of the study population, both for the entire population and by HbA1c group, are shown in Table 1. Systolic blood pressure and diastolic blood pressure increased with the increase in HbA1c level, whereas HDL-C level decreased. The increase in HbA1c level was also positively associated with dyslipidemia, unstable angina, and creatine kinase level. Table 2 summarizes the baseline clinical and laboratory characteristics of the patients according to the primary end points. Group 1 had 44 cases (9.9%), group 2 had 56 cases (14.7%), group 3 had 56 cases (14.9%), and group 4 had 41 cases (24.6%). Obviously, the incidence rate of the primary end points gradually increased with the increase of HbA1c level. Patients who experienced the primary end points more often had a previous MI. The perioperative period and surgical outcomes of the study population stratified by the primary end points are shown in Table 2. There is a certain difference in the rate of use of drug-coated balloons and drug-coated stents in patients with and without end point events.
Baseline Clinical Characteristics of Patients Stratified by the Optimal Cutoff Point of Glycated Hemoglobin (HbA1c) Level.
| Variable | Total population (n=1367) | Group 1 (n=444) | Group 2 (n=381) | Group 3 (n=375) | Group 4 (n=167) | P |
|---|---|---|---|---|---|---|
| Age, years | 58.8±10.3 | 58.2±10.7 | 58.8±10.1 | 59.7±10.0 | 58.2±10.4 | 0.169 |
| Male sex, n (%) | 979 (71.6) | 318 (71.6) | 286 (75.1) | 258 (68.8) | 117 (70.1) | 0.273 |
| BMI, kg/m2 | 26.0±3.3 | 25.9±3.2 | 25.9±3.3 | 26.1±3.5 | 26.2±3.1 | 0.753 |
| SBP, mmHg | 129.2±17.1 | 127.4±17.0 | 130.3±17.0 | 129.1±17.1 | 131.4±17.2 | 0.028 |
| DBP, mmHg | 77.3±11.0 | 77.3±11.0 | 78.2±10.7 | 76.2±11.2 | 77.9±10.9 | 0.076 |
| Smoker, n (%) | 698 (51.1) | 238 (53.6) | 194 (50.9) | 183 (48.8) | 83 (49.7) | 0.564 |
| Drinker, n (%) | 484 (35.4) | 182 (41.0) | 135 (35.4) | 121 (32.3) | 46 (27.5) | 0.007 |
| Medical history | ||||||
| DAPT, n (%) | 638 (46.7) | 220 (49.5) | 183 (48.0) | 154 (41.1) | 81 (48.5) | 0.081 |
| Hypertension, n (%) | 833 (60.9) | 254 (57.2) | 222 (58.3) | 247 (65.9) | 110 (65.9) | 0.026 |
| Dyslipidemia, n (%) | 992 (72.6) | 308 (69.4) | 273 (71.7) | 286 (76.3) | 125 (74.9) | 0.141 |
| Past PCI, n (%) | 330 (24.1) | 111 (25.0) | 86 (22.6) | 89 (23.7) | 44 (26.3) | 0.760 |
| Past CABG, n (%) | 21 (1.5) | 7 (1.6) | 5 (1.3) | 9 (2.4) | 0 (0) | 0.205 |
| Past MI, n (%) | 142 (10.4) | 44 (9.9) | 42 (11.0) | 37 (9.9) | 19 (11.4) | 0.906 |
| Clinical presentation | ||||||
| UA, n (%) | 1143 (83.6) | 351 (79.1) | 325 (85.3) | 326 (86.9) | 141 (84.4) | 0.014 |
| NSTEMI, n (%) | 68 (5.0) | 29 (6.5) | 12 (3.1) | 17 (4.5) | 10 (6.0) | 0.140 |
| STEMI, n (%) | 62 (4.5) | 28 (6.3) | 17 (4.5) | 11 (2.9) | 6 (3.6) | 0.122 |
| Laboratory results | ||||||
| TGs, mg/dL | 144.0±110.1 | 145. 8±113.5 | 143.8±112.6 | 144.6±101.8 | 137.9±113.8 | 0.888 |
| TC, mg/dL | 161.1±40.2 | 160.2±39.6 | 163.1±41.0 | 163.1±39.9 | 154.7±40.3 | 0.096 |
| LDL-C, mmol/L | 2.5±0.9 | 2.4±0.9 | 2.5±0.9 | 2.5±0.9 | 2.4±0.8 | 0.247 |
| HDL-C, mmol/L | 1.1±0.3 | 1.1±0.3 | 1.2±0.3 | 1.2±0.3 | 1.1±0.2 | 0.088 |
| CK, U/L | 123.9±214.7 | 113.5±139.2 | 158.5±343.0 | 109.6±108.5 | 105.7±175.5 | 0.005 |
| CK-MB, ng/mL | 5.2±22.0 | 5.5±26.3 | 5.5±20.7 | 5.6±19.8 | 2.5±4.6 | 0.775 |
| hs-CRP, mg/L | 3.0±5.2 | 3.5±5.8 | 2.3±4.0 | 3.0±5.3 | 2.8±5.4 | 0.248 |
| Creatinine, μmol/L | 71.5±15.0 | 70.7±14.1 | 70.8±15.2 | 72.9±16.0 | 72.3±14.5 | 0.126 |
| eGFR, mL/(min·1.73 m2) | 96.0±13.2 | 96.1±13.1 | 96.8±13.4 | 94.8±13.5 | 96.8±12.2 | 0.182 |
| Uric acid, μmol/L | 353.9±88.2 | 353.6±91.7 | 350.5±83.9 | 356.3±88.0 | 357.3±89.5 | 0.776 |
| FBG, mg/dL | 107.3±24.7 | 107.0±24.7 | 107.5±24.5 | 108.7±25.7 | 104.3±23.1 | 0.295 |
| HbA1c, % | 6.0±0.2 | 5.7±0.1 | 5.9±0.0 | 6.1±0.1 | 6.3±0.0 | <0.001 |
| Procedural results | ||||||
| DES implantation, n (%) | 631 (46.2) | 200 (45.0) | 183 (48.0) | 161 (42.9) | 87 (52.1) | 0.195 |
| DCB use, n (%) | 724 (53.0) | 231 (52.0) | 210 (55.1) | 186 (49.6) | 97 (58.1) | 0.228 |
| Number of stents | 0.8±1.0 | 0.7±1.0 | 0.8±1.0 | 0.8±1.1 | 0.9±1.0 | 0.534 |
BMI, body mass index; SBP, systolic blood pressure; DBP, diastolic blood pressure; DAPT, dual antiplatelet therapy; PCI, percutaneous coronary intervention; CABG, coronary artery bypass graft; MI, myocardial infarction; UA, unstable angina; NSTEMI, non–ST-segment elevation myocardial infarction; STEMI, ST-segment elevation myocardial infarction; TGs, triglycerides; TC, total cholesterol; LDL-C, low-density lipoprotein cholesterol; HDL-C, high-density lipoprotein cholesterol; CK, creatine kinase; CK-MB, creatine kinase myocardial band; hs-CRP, high-sensitivity C-reactive protein; eGFR, estimated glomerular filtration rate; FBG, fasting blood glucose; DES, drug-eluting stent; DCB, drug-coated balloon.
Baseline Clinical Characteristics of Patients if an Adverse Event Happened or if No Adverse Event Happened.
| Variable | Total population (n=1367) | No event (n=1170) | Primary end point (n=197) | P |
|---|---|---|---|---|
| Age, years | 58.8±10.3 | 58.9±10.4 | 58.5±10.1 | 0.651 |
| Male sex, n (%) | 979 (71.6) | 843 (72.1) | 136 (69.0) | 0.385 |
| BMI, kg/m2 | 26.0±3.3 | 26.0±3.2 | 25.7±3.6 | 0.232 |
| SBP, mmHg | 129.2±17.1 | 129.0±17.2 | 130.0±16.5 | 0.453 |
| DBP, mmHg | 77.3±11.0 | 77.4±10.9 | 77.1±11.7 | 0.780 |
| Smoker, n (%) | 698 (51.1) | 600 (51.3) | 98 (49.7) | 0.690 |
| Drinker, n (%) | 484 (35.4) | 420 (35.9) | 64 (32.5) | 0.354 |
| Medical history | ||||
| DAPT, n (%) | 638 (46.7) | 535 (45.7) | 103 (52.3) | 0.088 |
| Hypertension, n (%) | 833 (60.9) | 714 (61.0) | 119 (60.4) | 0.869 |
| Dyslipidemia, n (%) | 992 (72.6) | 861 (73.6) | 131 (66.5) | 0.039 |
| Past PCI, n (%) | 330 (24.1) | 280 (23.9) | 50 (25.4) | 0.660 |
| Past CABG, n (%) | 21 (1.5) | 19 (1.6) | 2 (1.0) | 0.756 |
| Past MI, n (%) | 142 (10.4) | 108 (9.2) | 34 (17.3) | 0.001 |
| Clinical presentation | ||||
| UA, n (%) | 1143 (83.6) | 979 (83.7) | 164 (83.2) | 0.881 |
| NSTEMI, n (%) | 68 (5.0) | 59 (5.0) | 9 (4.6) | 0.777 |
| STEMI, n (%) | 62 (4.5) | 53 (4.5) | 9 (4.6) | 0.981 |
| Laboratory results | ||||
| TGs, mg/dL | 144.0±110.1 | 144.4±112.9 | 141.4±91.9 | 0.726 |
| TC, mg/dL | 161.1±40.2 | 160.7±39.7 | 163.4±43.2 | 0.400 |
| LDL-C, mmol/L | 2.5±0.9 | 2.5±0.9 | 2.5±0.9 | 0.301 |
| HDL-C, mmol/L | 1.1±0.3 | 1.1±0.3 | 1.1±0.3 | 0.886 |
| CK, U/L | 123.9±214.7 | 126.6±227.7 | 107.4±109.4 | 0.249 |
| CK-MB, mg/mL | 5.2±22.0 | 4.5±20.1 | 9.5±31.6 | 0.841 |
| hs-CRP, mg/L | 3.0±5.2 | 2.9±5.1 | 3.2±5.7 | 0.467 |
| Creatinine, μmol/L | 71.5±15.0 | 71.7±15.1 | 70.6±14.3 | 0.302 |
| eGFR, mL/(min·1.73 m2) | 96.0±13.2 | 95.9±13.2 | 96.9±12.9 | 0.302 |
| Uric acid, μmol/L | 353.9±88.2 | 355.8±88.3 | 342.8±87.4 | 0.057 |
| FBG, mg/dL | 107.3±24.7 | 107.5±24.9 | 105.7±23.5 | 0.329 |
| HbA1c, % | 6.0±0.2 | 6.0±0.2 | 6.0±0.2 | <0.001 |
| Procedural results | ||||
| DES implantation, n (%) | 631 (46.2) | 528 (45.1) | 103 (52.3) | 0.062 |
| DCB use, n (%) | 724 (53.0) | 603 (51.5) | 121 (61.4) | 0.010 |
| Number of stents | 0.8±1.0 | 0.8±1.0 | 0.9±1.1 | 0.023 |
| HbA1c group | ||||
| 1, n (%) | 444 (32.5) | 400 (34.2) | 44 (22.3) | <0.001 |
| 2, n (%) | 381 (27.9) | 325 (27.8) | 56 (28.4) | – |
| 3, n (%) | 375 (27.4) | 319 (27.3) | 56 (28.4) | – |
| 4, n (%) | 167 (12.2) | 126 (10.8) | 41 (20.8) | – |
BMI, body mass index; SBP, systolic blood pressure; DBP, diastolic blood pressure; DAPT, dual antiplatelet therapy; PCI, percutaneous coronary intervention; CABG, coronary artery bypass graft; MI, myocardial infarction; UA, unstable angina; NSTEMI, non–ST-segment elevation myocardial infarction; STEMI, ST-segment elevation myocardial infarction; TGs, triglycerides; TC, total cholesterol; LDL-C, low-density lipoprotein cholesterol; HDL-C, high-density lipoprotein cholesterol; CK, creatine kinase; CK-MB, creatine kinase myocardial band; hs-CRP, high-sensitivity C-reactive protein; eGFR, estimated glomerular filtration rate; FBG, fasting blood glucose; HbA1c, glycated hemoglobin; DES, drug-eluting stent; DCB, drug-coated balloon.
Clinical Results and Kaplan-Meier analysis
The Kaplan-Meier curves of the incidence of the primary end point and the incidence of each constituent event of the primary end points across HbA1c groups are displayed in Figure 2. The incidence of the end point events gradually increased with the increase in HbA1c level. At all the end point events, nonfatal MI incidence increased by group; the rehospitalization rate for angina pectoris in groups 2, 3, and 4 was significantly higher than in group 1; and the rate of IDR in groups 2, 3, and 4 was also significantly higher than in group 1. On incorporation of traditional cardiovascular risk factors, such as male sex, age, BMI, smoker, drinker, hypertension, and hyperlipidemia, together with the risk factors for the end point events, Spearman or Pearson correlation analysis showed that the end point events were associated with a history of PCI, a history of MI, higher triglyceride level, and higher cholesterol levels. Higher LDL-C level, high-sensitivity C-reactive protein level, and eGFR were also associated with a higher risk of the end point events. In contrast, higher HDL-C and uric acid levels correlated with a lower rate of end point events. Interestingly, the use and quantity of drug-coated stents and the use of a drug-coated balloon had significant effects on the occurrence of end point events. In the univariate analysis, HbA1c level as a continuous variable showed an HR of 3.987 (95% CI 2.041–6.722; P<0.001) (Table 3); after adjustment for multiple confounding factors, the relationship remained significant (HR 4.110, 95% CI 2.097–6.011; P<0.001). Subsequent multivariate analysis of the primary end point events with group 1 as a reference category showed that the HbA1c level of groups 2, 3, and 4 was associated with the number of primary end point and other events (group 2, HR 1.624, 95% CI 1.057–2.495; group 3, HR 1.719, 95% CI 1.115–2.650; group 4, HR 3.111, 95% CI 1.918–5.046; Table 4). Indeed, after adjustment for age, male sex, BMI, previous MI, smoker status, drinker status, hypertension, dyslipidemia, uric acid level, stent implantation, use of a drug-coated balloon, and number of stents, HbA1c level still independently predicted the occurrence of MACE in patients with prediabetes (Figure 3).

Glycated Hemoglobin (HbA1c) Level and Risk: Kaplan-Meier Curves for Primary end Point Incidence (A), Nonfatal Myocardial Infarction (MI) Incidence (B), Incidence of Hospitalization for Unstable Angina (C), and Incidence of Ischemia-Driven Revascularization (D) for the Four Study Groups Based on HbA1c Level.
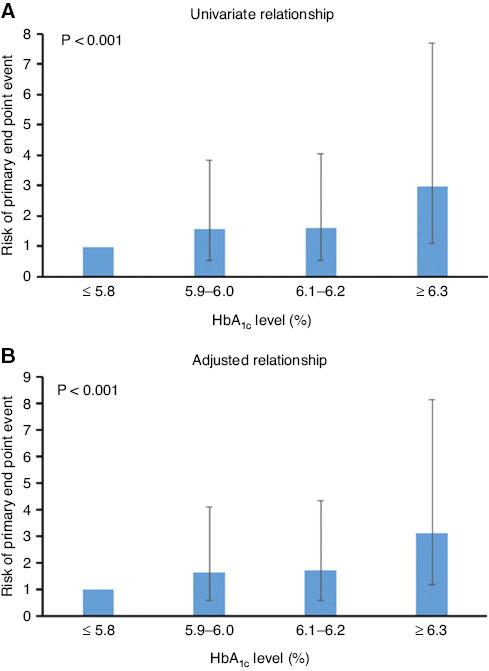
Risk of Nonfatal Myocardial Infarction, Hospitalization for Unstable Angina, or Ischemia-Driven Revascularization According to the Glycated Hemoglobin (HbA1c) Level.
The error bar represents a 95% confidence interval. (A) Univariate relationship. (B) Relationship adjusted for age, male sex, body mass index, previous myocardial infarction, smoker, drinker, hypertension, hyperlipidemia, stent implantation, balloon use, number of stents, and uric acid level.
Univariate and Multivariate Cox Regression Analysis for Predicting Major Adverse Cardiovascular Events.
| HR | Univariate 95% CI | P | HR | Multivariate 95% CI | P | |
|---|---|---|---|---|---|---|
| HbA1c level as a continuous variable | 3.987 | 2.041–6.722 | <0.001 | 4.110 | 2.097–6.011 | <0.001 |
| Age | 0.997 | 0.984–1.010 | 0.656 | |||
| Male sex | 0.864 | 0.639–1.168 | 0.347 | |||
| BMI | 0.973 | 0.932–1.016 | 0.218 | |||
| Smoker | 0.937 | 0.708–1.238 | 0.646 | |||
| Drinker | 0.861 | 0.639–1.160 | 0.320 | |||
| Hypertension | 0.969 | 0.728–1.289 | 0.828 | |||
| Dyslipidemia | 0.743 | 0.552–0.998 | 0.053 | |||
| Past PCI | 1.057 | 0.767–1.456 | 0.738 | |||
| Past CABG | 0.624 | 0.155–2.512 | 0.472 | |||
| Past MI | 1.860 | 1.285–2.691 | 0.002 | 1.928 | 1.327–2.803 | 0.001 |
| TGs | 1.000 | 0.998–1.001 | 0.700 | |||
| TC | 1.002 | 0.998–1.005 | 0.373 | |||
| LDL-C | 1.092 | 0.934–1.276 | 0.275 | |||
| HDL-C | 0.976 | 0.583–1.632 | 0.925 | |||
| CK | 0.999 | 0.998–1.001 | 0.180 | |||
| CK-MB | 1.006 | 1.000–1.011 | 0.980 | |||
| hs-CRP | 1.009 | 0.984–1.035 | 0.493 | |||
| Creatinine | 0.995 | 0.985–1.004 | 0.287 | |||
| eGFR | 1.006 | 0.995–1.017 | 0.292 | |||
| Uric acid | 0.998 | 0.997–1.000 | 0.047 | 0.998 | 0.996–1.000 | 0.016 |
| DES implantation | 1.313 | 0.993–1.737 | 0.056 | 0.764 | 0.419–1.394 | 0.380 |
| DCB use | 1.472 | 1.105–1.960 | 0.008 | 1.605 | 0.946–2.724 | 0.080 |
| Number of stents | 1.164 | 1.027–1.319 | 0.022 | 0.764 | 0.905–1.356 | 0.321 |
HR, hazard ratio; CI, confidence interval; HbA1c, glycated hemoglobin; BMI, body mass index; PCI, percutaneous coronary intervention; CABG, coronary artery bypass graft; MI, myocardial infarction; TGs, triglycerides; TC, total cholesterol; LDL-C, low-density lipoprotein cholesterol; HDL-C, high-density lipoprotein cholesterol; CK, creatine kinase; CK-MB, creatine kinase myocardial band; hs-CRP, high-sensitivity C-reactive protein; eGFR, estimated glomerular filtration rate; DES, drug-eluting stent; DCB, drug-coated balloon.
Baseline Glycated Hemoglobin (HbA1c) Level and Prediction of Cardiovascular Events.
| HbA1c group | Unadjusted HR (95% CI) | P | Adjusted HR (95% CI) | P | |
|---|---|---|---|---|---|
| Primary end point, n (%) | 1 | Reference | <0.001 | Reference | <0.001 |
| 2 | 1.566 (1.028–2.287) | 1.624 (1.057–2.495) | |||
| 3 | 1.596 (1.047–2.432) | 1.719 (1.115–2.650) | |||
| 4 | 2.958 (1.848–4.734) | 3.111 (1.918–5.046) | |||
| Nonfatal MI, n (%) | 1 | Reference | 0.470 | Reference | 0.476 |
| 2 | 2.939 (0.567–5.235) | 3.125 (0.589–6.562) | |||
| 3 | 2.986 (0.576–5.483) | 3.254 (0.607–6.640) | |||
| 4 | 4.043 (0.669–5.412) | 3.882 (0.610–6.709) | |||
| Hospitalization for unstable angina, n (%) | 1 | Reference | <0.001 | Reference | <0.001 |
| 2 | 1.437 (0.937–2.203) | 1.479 (0.958–2.286) | |||
| 3 | 1.496 (0.977–2.291) | 1.602 (1.035–2.480) | |||
| 4 | 2.863 (1.785–4.592) | 2.977 (1.832–4.836) | |||
| Ischemia-driven revascularization, n (%) | 1 | Reference | <0.001 | Reference | <0.001 |
| 2 | 1.173 (0.590–2.331) | 1.262 (0.625–2.548) | |||
| 3 | 1.871 (0.999–3.505) | 2.146 (1.121–4.107) | |||
| 4 | 5.500 (2.943–6.279) | 5.911 (3.081–6.338) |
The adjusted variables were male sex, age, body mass index, previous myocardial infarction (MI), smoker, drinker, hypertension, hyperlipidemia, stent implantation, balloon use, number of stents, and uric acid level.
HR, hazard ratio; CI, confidential interval.
Discussion
In the present study in individuals with CAD and prediabetes, we found a clear relationship between HbA1c level and adverse cardiovascular outcomes. Indeed, after adjustment for as many confounding factors and risk factors as possible, there was still an independent association between HbA1c level and negative outcomes. Subsequently, we conducted a clinical investigation to explore the relationship between HbA1c level and the incidence of CAD and mortality in patients with prediabetes and those without prediabetes.
HbA1c Level, Prediabetes, and Adverse Cardiovascular Events
Prediabetes and CAD
A number of studies have shown that prediabetes can lead to an increased risk of cardiovascular diseases. It has been shown that patients with prediabetes have an increased risk of T2DM and cardiovascular disease [12]. In a study involving 14,231 Chinese participants, Liu et al. [23] showed that the reversion of prediabetes to normal blood glucose levels reduced the risk of cardiovascular disease, MI, stroke, and all-cause death compared with progression to diabetes mellitus. A study in 5202 consecutive patients without diabetes mellitus who underwent PCI showed that the incidence of MACE in the prediabetes group was significantly greater than that in the nondiabetic group [24]. Zhao et al. [25] found that patients with prediabetes were at higher risk of developing diabetes mellitus during the follow-up. After adjustment for the severity of CAD, the risk of acute MI in patients with prediabetes was significantly higher. In contrast with individuals with normal blood glucose levels, patients with prediabetes after PCI had an increased risk of acute MI.
HbA1c Level, Prediabetes, and CAD
It has been shown that prediabetes increases the morbidity and mortality associated with cardiovascular diseases, so it is important to detect prediabetes at an early stage. Although the gold standard for diagnosing diabetes mellitus is the oral glucose tolerance test, HbA1c level is easy to obtain and is not affected by diet and stress. In addition, there are a growing number of studies on the relationship between HbA1c level and cardiovascular diseases. According to data from the New Zealand community, circulatory complications in 31,148 adults showed a strong correlation with HbA1c levels [26]. A study by Hu et al. [27] was based on repeated measurements of FBG and HbA1c levels; they found that prediabetes was closely related to a higher risk of cardiovascular disease. The United Kingdom Prospective Diabetes Study (UKPDS) 35 studied 3642 patients with newly diagnosed T2DM and found that HbA1c level was reduced by 1 percentage point and the relative risk of adverse cardiovascular events was reduced to various degrees (each 1% reduction in HbA1c was associated with 14%, 12%, and 16% reductions in the relative risk (RR) of myocardial infarction, stroke, and heart failure, respectively.) [28]. This conclusion is consistent with our study. With the increase in HbA1c level, adverse cardiovascular events occurred more often in patients with CAD with prediabetes.
HbA1c Level for Prognosis in Patients with Prediabetes and CAD
Raised HbA1c Level is a Risk Factor for Prediabetes with CAD
Raised HbA1c level is a significant risk factor for prediabetes or CAD. In the EPIC-Norfolk study, 10-year cardiovascular mortality was increased when HbA1c level within the normal range increased by 1 percentage point [29]. This phenomenon may be due to the toxic pathway activated by protein kinase C (PKC) that links the increase in blood glucose level with downstream mechanisms, leading to the expression of nitric oxide synthase, vascular endothelial growth factor, and plasminogen activator inhibitor 1. The downstream effects of PKC-related blood glucose level elevation, especially those involving the effects on blood vessels and inflammatory pathways, can explain the deterioration of B-cell function, insulin resistance, microvessels, and large blood vessels.
HbA1c May Have No Significant Effect
Some studies have shown that there is no correlation between prediabetes, HbA1c level, and the prognosis of cardiovascular disease. There was no serious association between HbA1c levels and cardiovascular prognosis in Korean individuals with prediabetes with ST-segment elevation MI (STEMI) who received direct PCI treatment [30]. That study involved 2470 STEMI patients after primary PCI who did not have diabetes mellitus (HbA1c level < 6.5% at admission). According to the HbA1c level at admission, the patients were divided into a prediabetes group (5.7% ≤ HbA1c level ≤ 6.4%) and a nondiabetic group (HbA1c level < 5.7%). It was found that HbA1c level at the time of hospitalization did not affect the occurrence of MACE. It may be that high levels of HbA1c were the result of long-term insulin resistance, which is the core of the pathophysiology of cardiometabolic syndrome, leading to dyslipidemia, hypercoagulable inflammation, and the incidence of cardiovascular events [31, 32].
The Positive Effects of Early Intervention in Prediabetes
Previous studies have shown that prediabetes worsens prognosis in patients with CAD, and for patients with CAD it may be beneficial to incorporate prediabetes evaluation and for them to undergo a long-term management program. A cost-effective way to prevent diabetes mellitus is lifestyle intervention, which has been shown to reduce the relative risk of T2DM by 40–70% [33–35] and also decrease cardiovascular and all-cause mortality and complications of diabetes mellitus [33]. In addition to lifestyle interventions, some drugs may prevent diabetes mellitus in patients with prediabetes, while others have been shown to delay or avert the transition from prediabetes to T2DM [36–38]. In view of the increasing burden of diabetes mellitus worldwide, it is necessary to advocate the screening and identification of patients with prediabetes. On the basis of the results of our study, strengthening the monitoring and control of HbA1c level in patients with prediabetes complicated with CAD can effectively improve the prognosis.
Limitations
Our research has some limitations. First and foremost, it was a retrospective observational analysis from a single center, with a small sample size; thus, to validate the results of this study, multicenter studies are needed. Second, the severity of CAD was not graded. It would be useful to use the SYNTAX score to evaluate the severity of CAD. Third, we assessed HbA1c level only once, at admission, and did not monitor its changes during the follow-up. In subsequent follow-ups, patients could be scheduled to undergo HbA1c level measurement on a regular basis, so as to obtain dynamic follow-up results. Fourth, the current research results included only Chinese patients. Considering the different ethnic metabolic levels, these results should be further verified through multicenter studies in multiple countries and regions, including different ethnic groups. Fifth, dormant malignancies and other undiagnosed systemic diseases were not ruled out, and these may have had an impact on the prognosis. During the follow-up, we tried to exclude patients with tumors and systemic diseases to minimize their impact on the results of the study.
Conclusions
HbA1c level is significantly associated with the risk of MACE in Asian patients with prediabetes and CAD. Even after adjustment for other confounding factors, the HbA1c level had a significant prognostic value in the prediction of MACE. Moreover, HbA1c level is stabler than FBG level. Taken together, for Asian patients with prediabetes and CAD, HbA1c level is a valuable marker for the prognosis of cardiovascular events.

