- Record: found
- Abstract: found
- Article: found
UCP2 KO mice exhibit ameliorated obesity and inflammation induced by high-fat diet feeding

Read this article at
Abstract
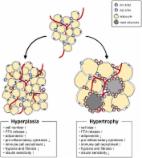
Uncoupling protein 2 (Ucp2) was first introduced as a member of Uncoupling protein family and a regulator of ROS formation; however, its role in adipose tissue is not fully understood. In the present study, we have investigated the role of Ucp2 against high-fat diet (HFD)-induced obesity in epididymal white adipose tissue (eWAT) and browning of inguinal white adipose tissue (iWAT). Diet-induced obesity is closely related to macrophage infiltration and the secretion of pro-inflammatory cytokines. Macrophages surround adipocytes and form a crown-like-structure (CLS). Some reports have suggested that CLS formation requires adipocyte apoptosis. After 12 weeks of HFD challenge, Ucp2 knockout (KO) mice maintained relatively lean phenotypes compared to wild-type (WT) mice. In eWAT, macrophage infiltration, CLS formation, and inflammatory cytokines were reduced in HFD KO mice compared to HFD WT mice. Surprisingly, we found that apoptotic signals were also reduced in the Ucp2 KO mice. Our study suggests that Ucp2 deficiency may prevent diet-induced obesity by regulating adipocyte apoptosis. However, Ucp2 deficiency did not affect the browning capacity of iWAT.
Related collections
Most cited references34
- Record: found
- Abstract: not found
- Article: not found
Obesity is associated with macrophage accumulation in adipose tissue
- Record: found
- Abstract: found
- Article: found
Adipose Tissue Remodeling: Its Role in Energy Metabolism and Metabolic Disorders
- Record: found
- Abstract: found
- Article: not found
Disruption of the uncoupling protein-2 gene in mice reveals a role in immunity and reactive oxygen species production.
Author and article information
Comments
Comment on this article
 Smart Citations
Smart CitationsSee how this article has been cited at scite.ai
scite shows how a scientific paper has been cited by providing the context of the citation, a classification describing whether it supports, mentions, or contrasts the cited claim, and a label indicating in which section the citation was made.