- Record: found
- Abstract: found
- Article: not found
Exosomes Secreted by Human-Induced Pluripotent Stem Cell-Derived Mesenchymal Stem Cells Repair Critical-Sized Bone Defects through Enhanced Angiogenesis and Osteogenesis in Osteoporotic Rats
Read this article at
Abstract
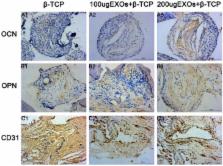
Bone defects caused by trauma, severe infection, tumor resection and skeletal abnormalities are common osteoporotic conditions and major challenges in orthopedic surgery, and there is still no effective solution to this problem. Consequently, new treatments are needed to develop regeneration procedures without side effects. Exosomes secreted by mesenchymal stem cells (MSCs) derived from human induced pluripotent stem cells (hiPSCs, hiPSC-MSC-Exos) incorporate the advantages of both MSCs and iPSCs with no immunogenicity. However, there are no reports on the application of hiPSC-MSC-Exos to enhance angiogenesis and osteogenesis under osteoporotic conditions. HiPSC-MSC-Exos were isolated and identified before use. The effect of hiPSC-MSC-Exos on the proliferation and osteogenic differentiation of bone marrow MSCs derived from ovariectomized (OVX) rats (rBMSCs-OVX) in vitro were investigated. In vivo, hiPSC-MSC-Exos were implanted into critical size bone defects in ovariectomized rats, and bone regeneration and angiogenesis were examined by microcomputed tomography (micro-CT), sequential fluorescent labeling analysis, microfil perfusion and histological and immunohistochemical analysis. The results in vitro showed that hiPSC-MSC-Exos enhanced cell proliferation and alkaline phosphatase (ALP) activity, and up-regulated mRNA and protein expression of osteoblast-related genes in rBMSCs-OVX. In vivo experiments revealed that hiPSC-MSC-Exos dramatically stimulated bone regeneration and angiogenesis in critical-sized calvarial defects in ovariectomized rats. The effect of hiPSC-MSC-Exos increased with increasing concentration. In this study, we showed that hiPSC-MSC-Exos effectively stimulate the proliferation and osteogenic differentiation of rBMSCs-OVX, with the effect increasing with increasing exosome concentration. Further analysis demonstrated that the application of hiPSC-MSC-Exos+β-TCP scaffolds promoted bone regeneration in critical-sized calvarial defects by enhancing angiogenesis and osteogenesis in an ovariectomized rat model.
Related collections
Most cited references48
- Record: found
- Abstract: found
- Article: not found
Osteoporosis: now and the future.
- Record: found
- Abstract: found
- Article: not found
Reprogramming of human somatic cells to pluripotency with defined factors.

- Record: found
- Abstract: found
- Article: found
