- Record: found
- Abstract: found
- Article: found
In Silico Drug Design for Purinergic GPCRs: Overview on Molecular Dynamics Applied to Adenosine and P2Y Receptors
Read this article at
Abstract
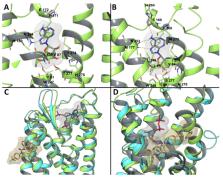
Molecular modeling has contributed to drug discovery for purinergic GPCRs, including adenosine receptors (ARs) and P2Y receptors (P2YRs). Experimental structures and homology modeling have proven to be useful in understanding and predicting structure activity relationships (SAR) of agonists and antagonists. This review provides an excursus on molecular dynamics (MD) simulations applied to ARs and P2YRs. The binding modes of newly synthesized A 1AR- and A 3AR-selective nucleoside derivatives, potentially of use against depression and inflammation, respectively, have been predicted to recapitulate their SAR and the species dependence of A 3AR affinity. P2Y 12R and P2Y 1R crystallographic structures, respectively, have provided a detailed understanding of the recognition of anti-inflammatory P2Y 14R antagonists and a large group of allosteric and orthosteric antagonists of P2Y 1R, an antithrombotic and neuroprotective target. MD of A 2AAR (an anticancer and neuroprotective target), A 3AR, and P2Y 1R has identified microswitches that are putatively involved in receptor activation. The approach pathways of different ligands toward A 2AAR and P2Y 1R binding sites have also been explored. A 1AR, A 2AAR, and A 3AR were utilizes to study allosteric phenomena, but locating the binding site of structurally diverse allosteric modulators, such as an A 3AR enhancer LUF6000, is challenging. Ligand residence time, a predictor of in vivo efficacy, and the structural role of water were investigated through A 2AAR MD simulations. Thus, new MD and other modeling algorithms have contributed to purinergic GPCR drug discovery.
Related collections
Most cited references125
- Record: found
- Abstract: found
- Article: not found
Adenosine receptors as therapeutic targets.
- Record: found
- Abstract: found
- Article: not found
Structural basis for allosteric regulation of GPCRs by sodium ions.
- Record: found
- Abstract: found
- Article: not found