- Record: found
- Abstract: found
- Article: found
Therapeutic Potential to Modify the Mucus Barrier in Inflammatory Bowel Disease
Read this article at
Abstract
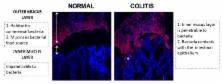
Recently, numerous studies have shown that disruption of the mucus barrier plays an important role in the exacerbation of inflammatory bowel disease, particularly in ulcerative colitis. Alterations in the mucus barrier are well supported by published data and are widely accepted. The use of fluorescence in situ hybridization and Carnoy’s fixation has revealed the importance of the mucus barrier in maintaining a mutualistic relationship between host and bacteria. Studies have raised the possibility that modulation of the mucus barrier may provide therapies for the disease, using agents such as short-chain fatty acids, prebiotics and probiotics. This review describes changes in the mucus barrier of patients with inflammatory bowel disease and in animal models of the disease. We also review the involvement of the mucus barrier in the exacerbation of the disease and explore the therapeutic potential of modifying the mucus barrier with short-chain fatty acids, prebiotics, probiotics, fatty acid synthase, H 2S, neutrophil elastase inhibitor and phophatidyl choline.
Related collections
Most cited references87
- Record: found
- Abstract: found
- Article: not found
The microbiome in inflammatory bowel disease: current status and the future ahead.
- Record: found
- Abstract: found
- Article: not found
The two mucus layers of colon are organized by the MUC2 mucin, whereas the outer layer is a legislator of host-microbial interactions.
- Record: found
- Abstract: found
- Article: not found