- Record: found
- Abstract: found
- Article: found
Four‐year results of a minimally invasive transscleral glaucoma gel stent implantation in a prospective multi‐centre study
Read this article at
Abstract
Importance
The transscleral XEN Glaucoma Gel Microstent (XEN‐GGM, Allergan Plc., Parsippany, New Jersey) is implanted by a minimally invasive ab interno technique.
Background
The present study aims to assess the long‐term clinical outcomes in patients after XEN‐GGM implantation.
Design
This prospective, non‐randomized, multi‐centred study was conducted in three countries (Austria, Canada and Germany).
Participants
Sixty‐four consecutive eyes of 64 patients with open angle glaucoma received the XEN‐GGM (63 μm) without Mitomycin C. Thirty‐five (55%) were solo procedures, and 29 (45%) were combined with cataract surgery.
Main Outcome Measures
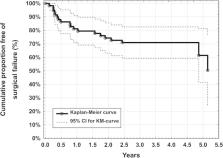
The main outcome measures were mean intraocular pressure (IOP), mean number of IOP lowering medication. Secondary outcome parameters were: visual acuity, visual fields and complete surgical failure (defined as presence of a secondary IOP lowering procedure or loss of light perception) at 4 years, postoperatively.
Results
Mean best‐medicated baseline IOP was 22.5 ± 4.2 mmHg and decreased significantly to 13.4 ± 3.1 mmHg 4 years postoperatively (−40%, n = 34, P < 0.001). Mean number of IOP lowering medication decreased significantly from 2.4 ± 1.3 preoperatively to 1.2 ± 1.3 (−50%, n = 34, P < 0.001) postoperatively. Visual field mean deviation showed no significant change between preoperative and postoperative examinations. Complete surgical failure rate per year was 10%.
Related collections
Most cited references24
- Record: found
- Abstract: not found
- Article: not found
Trabeculectomy. Preliminary report of a new method.

- Record: found
- Abstract: found
- Article: found
Minimally invasive glaucoma surgery: current status and future prospects
- Record: found
- Abstract: found
- Article: not found