- Record: found
- Abstract: found
- Article: found
The Extracellular Matrix Component Psl Provides Fast-Acting Antibiotic Defense in Pseudomonas aeruginosa Biofilms
Read this article at
Abstract
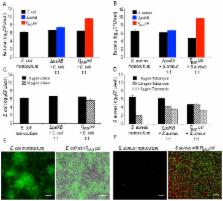
Bacteria within biofilms secrete and surround themselves with an extracellular matrix, which serves as a first line of defense against antibiotic attack. Polysaccharides constitute major elements of the biofilm matrix and are implied in surface adhesion and biofilm organization, but their contributions to the resistance properties of biofilms remain largely elusive. Using a combination of static and continuous-flow biofilm experiments we show that Psl, one major polysaccharide in the Pseudomonas aeruginosa biofilm matrix, provides a generic first line of defense toward antibiotics with diverse biochemical properties during the initial stages of biofilm development. Furthermore, we show with mixed-strain experiments that antibiotic-sensitive “non-producing” cells lacking Psl can gain tolerance by integrating into Psl-containing biofilms. However, non-producers dilute the protective capacity of the matrix and hence, excessive incorporation can result in the collapse of resistance of the entire community. Our data also reveal that Psl mediated protection is extendible to E. coli and S. aureus in co-culture biofilms. Together, our study shows that Psl represents a critical first bottleneck to the antibiotic attack of a biofilm community early in biofilm development.
Author Summary
Many bacteria have the ability to form multicellular communities, termed biofilms. An important characteristic of a biofilm is the ability of cells to synthesize and secrete an extracellular matrix. This matrix offers structural support, community organization, and added protection, often making the cells impervious to desiccation, predation, and antimicrobials. In this study, we investigate the contributions of polysaccharide components found in the extracellular matrix of Pseudomonas aeruginosa at progressive stages in biofilm development. We first show that one specific polysaccharide, Psl, provides an added defense for P. aeruginosa biofilms against antimicrobials of different properties for young biofilms. Then, by cultivating biofilms that contain both Psl producing and Psl non-producing strains, we find that P. aeruginosa, E. coli, and S. aureus species that lack Psl take advantage of the protection offered by cells producing Psl. Collectively, the data indicate that Psl is likely to play a key protective role in early development of P. aeruginosa biofilm associated infections.
Related collections
Most cited references47
- Record: found
- Abstract: found
- Article: not found
NIH Image to ImageJ: 25 years of image analysis.
- Record: found
- Abstract: found
- Article: not found
Colistin: the revival of polymyxins for the management of multidrug-resistant gram-negative bacterial infections.
- Record: found
- Abstract: found
- Article: not found