- Record: found
- Abstract: found
- Article: found
The C-terminal basic residues contribute to the chemical- and voltage-dependent activation of TRPA1
Read this article at
Abstract
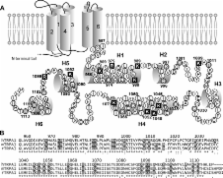
The ankyrin transient receptor potential channel TRPA1 is a non-selective cationic channel that is expressed by sensory neurons, where it can be activated by pungent chemicals, such as AITC (allyl isothiocyanate), cinnamon or allicin, by deep cooling (<18 °C) or highly depolarizing voltages (>+100 mV). From the cytoplasmic side, this channel can be regulated by negatively charged ligands such as phosphoinositides or inorganic polyphosphates, most likely through an interaction with as yet unidentified positively charged domain(s). In the present study, we mutated 27 basic residues along the C-terminal tail of TRPA1, trying to explore their role in AITC- and voltage-dependent gating. In the proximal part of the C-terminus, the function-affecting mutations were at Lys 969, Arg 975, Lys 988 and Lys 989. A second significant region was found in the predicted helix, centred around Lys 1048 and Lys 1052, in which single alanine mutations completely abolished AITC- and voltage-dependent activation. In the distal portion of the C-terminus, the charge neutralizations K1092A and R1099A reduced the AITC sensitivity, and, in the latter mutant, increased the voltage-induced steady-state responses. Taken together, our findings identify basic residues in the C-terminus that are strongly involved in TRPA1 voltage and chemical sensitivity, and some of them may represent possible interaction sites for negatively charged molecules that are generally considered to modulate TRPA1.
Related collections
Most cited references37
- Record: found
- Abstract: found
- Article: not found
TRPA1 mediates the inflammatory actions of environmental irritants and proalgesic agents.
- Record: found
- Abstract: found
- Article: not found
Noxious cold ion channel TRPA1 is activated by pungent compounds and bradykinin.
- Record: found
- Abstract: found
- Article: not found