- Record: found
- Abstract: found
- Article: not found
The Structural Basis of Gas-Responsive Transcription by the Human Nuclear Hormone Receptor REV-ERBβ
Read this article at
Abstract
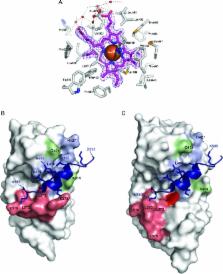
Heme is a ligand for the human nuclear receptors (NR) REV-ERBα and REV-ERBβ, which are transcriptional repressors that play important roles in circadian rhythm, lipid and glucose metabolism, and diseases such as diabetes, atherosclerosis, inflammation, and cancer. Here we show that transcription repression mediated by heme-bound REV-ERBs is reversed by the addition of nitric oxide (NO), and that the heme and NO effects are mediated by the C-terminal ligand-binding domain (LBD). A 1.9 Å crystal structure of the REV-ERBβ LBD, in complex with the oxidized Fe(III) form of heme, shows that heme binds in a prototypical NR ligand-binding pocket, where the heme iron is coordinately bound by histidine 568 and cysteine 384. Under reducing conditions, spectroscopic studies of the heme-REV-ERBβ complex reveal that the Fe(II) form of the LBD transitions between penta-coordinated and hexa-coordinated structural states, neither of which possess the Cys384 bond observed in the oxidized state. In addition, the Fe(II) LBD is also able to bind either NO or CO, revealing a total of at least six structural states of the protein. The binding of known co-repressors is shown to be highly dependent upon these various liganded states. REV-ERBs are thus highly dynamic receptors that are responsive not only to heme, but also to redox and gas. Taken together, these findings suggest new mechanisms for the systemic coordination of molecular clocks and metabolism. They also raise the possibility for gas-based therapies for the many disorders associated with REV-ERB biological functions.
Author Summary
Much of human biology, such as sleeping, eating, and even the prevalence of heart attacks, occurs in daily cycles. These cycles are orchestrated by a master “clock” located in the brain. The basic components of this clock are proteins that control the expression of important genes. In this study, we analyze one of these regulatory proteins, named REV-ERB, and show that it is regulated by the combination of heme and nitric oxide gas, both of which are important regulators of human physiology. By determining the 3-D structure of the REV-ERB protein, we were able to uncover clues as to how this regulation occurs. REV-ERB belongs to a protein family called nuclear hormone receptors, which are known to be excellent drug targets. Thus, this paper opens the door to possible gas-based therapies for diseases known to involve REV-ERB, such as diabetes, atherosclerosis, inflammation, and cancer.
Abstract
The heme-regulated nuclear hormone receptor REV-ERB is one of the core transcription factors regulating circadian rhythms. We found that transcription by heme-bound REV-ERB is regulated by NO gas, and a crystal structure of the heme-bound protein uncovers the basis for heme and gas binding.
Related collections
Most cited references91
- Record: found
- Abstract: found
- Article: not found
Multiple sequence alignment with the Clustal series of programs.
- Record: found
- Abstract: found
- Article: not found
Coordinated transcription of key pathways in the mouse by the circadian clock.
- Record: found
- Abstract: found
- Article: not found
