- Record: found
- Abstract: found
- Article: found
MicroRNA‑17‑5p alleviates sepsis‑related acute kidney injury in mice by modulating inflammation and apoptosis
Read this article at
Abstract
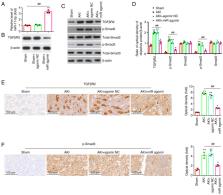
Septic acute kidney injury (AKI) is considered as a severe and frequent complication that occurs during sepsis. Mounting evidence has confirmed the pivotal pathogenetic roles of microRNA (miRNA or miR) in sepsis-induced AKI; however, the role of miRNAs and their underlying mechanisms in sepsis-induced AKI have not been entirely understood. The present study aimed to elucidate the functions of special miRNAs during sepsis-induced AKI and its underlying mechanism. First, a number of differently expressed miRNAs was identified based on the microarray dataset GSE172044. Subsequently, lipopolysaccharide (LPS) was used to induce AKI in mice, and the role of miR-17-5p on AKI was clarified. Finally, the related molecular mechanisms were further examined by western blotting and immunohistochemical analysis. MiR-17-5p was found to be continuously decreased and reached the bottom at h 24 after AKI in mice. Functionally, injection of agomiR-17-5p could observably improve renal injury and survival rate, as well as inhibit inflammatory cytokine production and renal cell apoptosis in mice after AKI. On the contrary, injection of antagomiR-17-5p aggravated LPS-induced renal injury, inflammation and apoptosis in mice after AKI. Moreover, transforming growth factor β receptor 2 (TGFβR2) was identified as a direct target of miR-17-5p, and its downstream phosphorylated Smad3 was also suppressed by miR-17-5p upregulation. Taken together, these results demonstrated that miR-17-5p overexpression may exhibit a beneficial effect by attenuating LPS-induced inflammation and apoptosis via regulating the TGFβR2/TGF-β/Smad3 signaling pathway, indicating that miR-17-5p could act as a potential target for sepsis treatment.
Related collections
Most cited references52
- Record: found
- Abstract: found
- Article: not found
Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) Method.

- Record: found
- Abstract: found
- Article: found
limma powers differential expression analyses for RNA-sequencing and microarray studies
- Record: found
- Abstract: found
- Article: not found