- Record: found
- Abstract: found
- Article: found
MAP1B Regulates Axonal Development by Modulating Rho-GTPase Rac1 Activity
Read this article at
Abstract
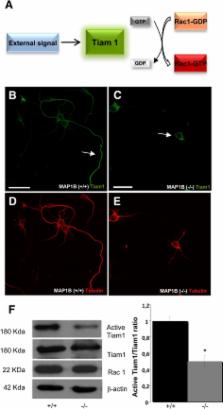
This article shows a novel function for the MAP1B protein, related to the control of actin dynamics through interaction with Tiam1.
Abstract
Cultured neurons obtained from MAP1B-deficient mice have a delay in axon outgrowth and a reduced rate of axonal elongation compared with neurons from wild-type mice. Here we show that MAP1B deficiency results in a significant decrease in Rac1 and cdc42 activity and a significant increase in Rho activity. We found that MAP1B interacted with Tiam1, a guanosine nucleotide exchange factor for Rac1. The decrease in Rac1/cdc42 activity was paralleled by decreases in the phosphorylation of the downstream effectors of these proteins, such as LIMK-1 and cofilin. The expression of a constitutively active form of Rac1, cdc42, or Tiam1 rescued the axon growth defect of MAP1B-deficient neurons. Taken together, these observations define a new and crucial function of MAP1B that we show to be required for efficient cross-talk between microtubules and the actin cytoskeleton during neuronal polarization.
Related collections
Most cited references40
- Record: found
- Abstract: found
- Article: not found
Signaling from Rho to the actin cytoskeleton through protein kinases ROCK and LIM-kinase.
- Record: found
- Abstract: found
- Article: not found
Cofilin phosphorylation by LIM-kinase 1 and its role in Rac-mediated actin reorganization.
- Record: found
- Abstract: found
- Article: not found