- Record: found
- Abstract: found
- Article: not found
Towards new horizons: characterization, classification and implications of the tumour antigenic repertoire
Read this article at
Abstract
Immune-checkpoint inhibition provides an unmatched level of durable clinical efficacy in various malignancies. Such therapies promote the activation of antigen-specific T cells, although the precise targets of these T cells remain unknown. Exploiting these targets holds great potential to amplify responses to treatment, such as by combining immune-checkpoint inhibition with therapeutic vaccination or other antigen-directed treatments. In this scenario, the pivotal hurdle remains the definition of valid HLA-restricted tumour antigens, which requires several levels of evidence before targets can be established with sufficient confidence. Suitable antigens might include tumour-specific antigens with alternative or wild-type sequences, tumour-associated antigens and cryptic antigens that exceed exome boundaries. Comprehensive antigen classification is required to enable future clinical development and the definition of innovative treatment strategies. Furthermore, clinical development remains challenging with regard to drug manufacturing and regulation, as well as treatment feasibility. Despite these challenges, treatments based on diligently curated antigens combined with a suitable therapeutic platform have the potential to enable optimal antitumour efficacy in patients, either as monotherapies or in combination with other established immunotherapies. In this Review, we summarize the current state-of-the-art approaches for the identification of candidate tumour antigens and provide a structured terminology based on their underlying characteristics.
Abstract
Immune-checkpoint inhibition has transformed the treatment of patients with advanced-stage cancers. Nonetheless, the specific antigens targeted by T cells that are activated or reactivated by these agents remain largely unknown. In this Review, the authors describe the characterization and classification of tumour antigens including descriptions of the most appropriate detection methods, and discuss potential regulatory issues regarding the use of tumour antigen-based therapeutics.
Key points
-
Immune-checkpoint inhibition has profoundly changed the paradigm for the care of several malignancies. Although these therapies activate antigen-specific T cells, the precise mechanisms of action and their specific targets remain largely unknown.
-
Anticancer immunotherapies encompass two fundamentally different therapeutic principles based on knowledge of their therapeutic targets, that either have been characterized (antigen-aware) or have remained elusive (antigen-unaware).
-
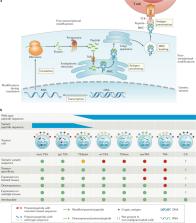
HLA-presented tumour antigens of potential therapeutic relevance can comprise alternative or wild-type amino acid sequences and can be subdivided into different categories based on their mechanisms of formation.
-
The available methods for the detection of HLA-presented antigens come with intrinsic challenges and limitations and, therefore, warrant multiple lines of evidence of robust tumour specificity before being considered for clinical use.
-
Knowledge obtained using various antigen-detection strategies can be combined with different therapeutic platforms to create individualized therapies that hold great promise, including when combined with already established immunotherapies.
-
Tailoring immunotherapies while taking into account the substantial heterogeneity of malignancies as well as that of HLA loci not only requires innovative science, but also demands innovative approaches to trial design and drug regulation.
Related collections
Most cited references120
- Record: found
- Abstract: found
- Article: not found
Neoantigen vaccine generates intratumoral T cell responses in phase Ib glioblastoma trial
- Record: found
- Abstract: found
- Article: not found
Predicting immunogenic tumour mutations by combining mass spectrometry and exome sequencing.
- Record: found
- Abstract: found
- Article: not found
